
Food Safety Focus (36th Issue, July 2009) – Food Safety Platform
Genetically Modified Food – Allergenicity and Safety Assessment
Reported by Ms. Shuk-man CHOW, Scientific Officer,
Risk Assessment Section,
Centre for Food Safety
After introducing the basics on genetically modified ( GM ) food last month, we are going to discuss, in this issue, the potential allergenicity and safety assessment of crops derived from genetic engineering.
Allergenicity of GM Crops
Allergenicity is the capacity to be an allergen that elicits a hypersensitive immune reaction. Almost all food allergens are proteins and the allergy-causing proteins tend to be relatively small, and resistant to heat, acid, and stomach enzyme degradation. While the crops used as staple foods contain tens of thousands of different proteins, relatively few are allergenic. However, the use of genetic engineering to modify the genetic make-up of crop plants might alter their allergenic potential.
Allergenic proteins may be introduced into newly developed GM crops if the transgene is from a source known to be allergenic. A well-known example is an experimental GM soyabean engineered to contain a Brazil nut (a known allergenic food) methionine-rich protein was found to cause allergic reaction in patients sensitive to this nut. As a result, the GM soyabean was withdrawn from further development and has never been marketed. As a measure to prevent the introduction of allergen to GM crops, the transfer of genes from commonly allergenic foods is discouraged.
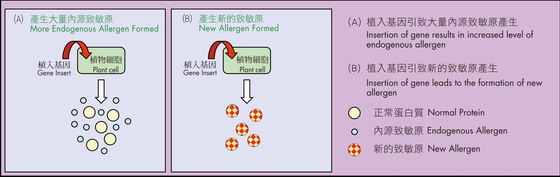
Due to the occurrence of unintended effects during the process of genetic engineering, transfer of genes from foods that are not commonly known to cause allergies can have the potential to cause allergic reaction. "Unintended effects" are the effects which go beyond that of the original modification and may arise through the random insertion of DNA sequence into the plant genome. The gene insert may cause modification in the expression of endogenous genes leading to either over-expression or under-expression of specific proteins. If the host plant contains known allergenic proteins, adding a new gene into it could increase the expression of the allergens and the plant may become more allergenic (Figure 1A). It is also possible that the insertion of the DNA sequence creates additional new proteins that are allergenic to susceptible individuals (Figure 1B).
Figure 1: Possible unintended allergenic effects
Safety Assessment of GM Food
Besides allergenicity, genetic modification might also alter the toxicity and chemical composition of a transgenic plant. Taking into account both intended and unintended changes that may occur due to genetic modification, the Codex Alimentarius Commission (Codex) has set out specific guidelines for safety assessment of food derived from GM organisms.
Under the assessment scheme, factors of health concerns such as the possible toxicity and allergenicity of the gene product, composition of key components, levels of various metabolites and the nutritional impact of the new GM crop will be evaluated and compared to the conventional counterpart. If no major difference is identified from the comparison, the newly constructed GM crop may be considered as safe as its conventional counterpart, or else, the impact on food safety attributable to the differences identified will be evaluated by additional assessment and animal studies.
This comparative analysis is regarded by many international authorities as the most appropriate strategy for safety assessment of GM food. National authorities of GM food producing countries have adopted the comparative approach and based on which to establish structured pre-market safety assessment scheme to evaluate the safety of food derived from genetic engineering. Any newly developed GM food should have undergone such assessment before approval for market sale. In case where potential hazard is identified from the assessment, the crop under evaluation should never be released for human consumption. GM food available in the international market has undergone safety assessment evaluation and is safe for human consumption.
Examples of GM crops approved for food use
| GM crops | Potential uses |
|---|---|
| Soyabean | Soy beverages, tofu, soy oil, soy flour, and as ingredients in breads, pastries and edible oil, etc |
| Corn | Corn oil, corn flour, sugar or syrup, and as ingredients in snacks, bakery, confectionery and soft drink, etc |
| Tomato | Tomato puree, tomato juice |
| Potato | Potato chips, mashed potato, potato soup, starch |
| Papaya | Papaya soup and desserts |
| Squash | Squash soup and desserts |
| Rice | Staple food, rice flour and noodle |
Glossary
| Gene | A gene is a unit of hereditary material, which carries the required information necessary to produce a protein(s) that determines the characteristics of an organism. |
|---|---|
| Genetic engineering | A technique which enables direct manipulation of genes of organisms. |
| Genetically modified food | Any food which is, or is derived from, genetically modified organisms. |
| Genome | A complete set of chromosomes found in nucleus of a given species which contains the entire genetic materials. |
| Transgene | The gene of interest to be introduced into the host organism. |


