
Food Safety Focus (205th Issue, August 2023) – Article 2
Aspartame: Safe or Not?
Reported by Ms. Sosanna WONG, Scientific Officer,
Risk Assessment Section, Centre for Food Safety
Aspartame has been the subject of extensive investigation for more than 40 years, and its safety has been comprehensively reviewed by international organisations such as the Joint FAO/WHO Expert Committee for Food Additives (JECFA), the European Food Safety Authority (EFSA), the US Food and Drug Administration (FDA) and Food Standards Australia New Zealand (FSANZ). These organisations have concluded unanimously that scientific evidence supports the safety of aspartame for use as a sweetener. Nonetheless, given the availability of new research results, the International Agency for Research on Cancer (IARC) and JECFA have recently conducted independent and complementary reviews to assess the potential carcinogenic hazard and other health risks associated with aspartame consumption.
What is Aspartame?
Aspartame is an intense, low-calorie artificial sweetener widely used since the 1980s in various food products like soda, chewing gum and ice-cream. It is approximately 200 times sweeter than table sugar and thus smaller amounts can achieve the same level of sweetness as sugar in food.
Aspartame consists of two amino acid moieties, aspartic acid and phenylalanine. The phenylalanine moiety has been slightly modified to give aspartame its sweet taste. After oral ingestion, aspartame is fully hydrolysed in the gastrointestinal tract to yield aspartic acid, phenylalanine and methanol, which are all occurring naturally in the body and in a wide variety of foods. These metabolites are then absorbed into the blood and are further metabolised. The two amino acids (i.e. aspartic acid and phenylalanine) from proteins in food or from aspartame are chemically indistinguishable and are metabolised in the same way in our bodies.
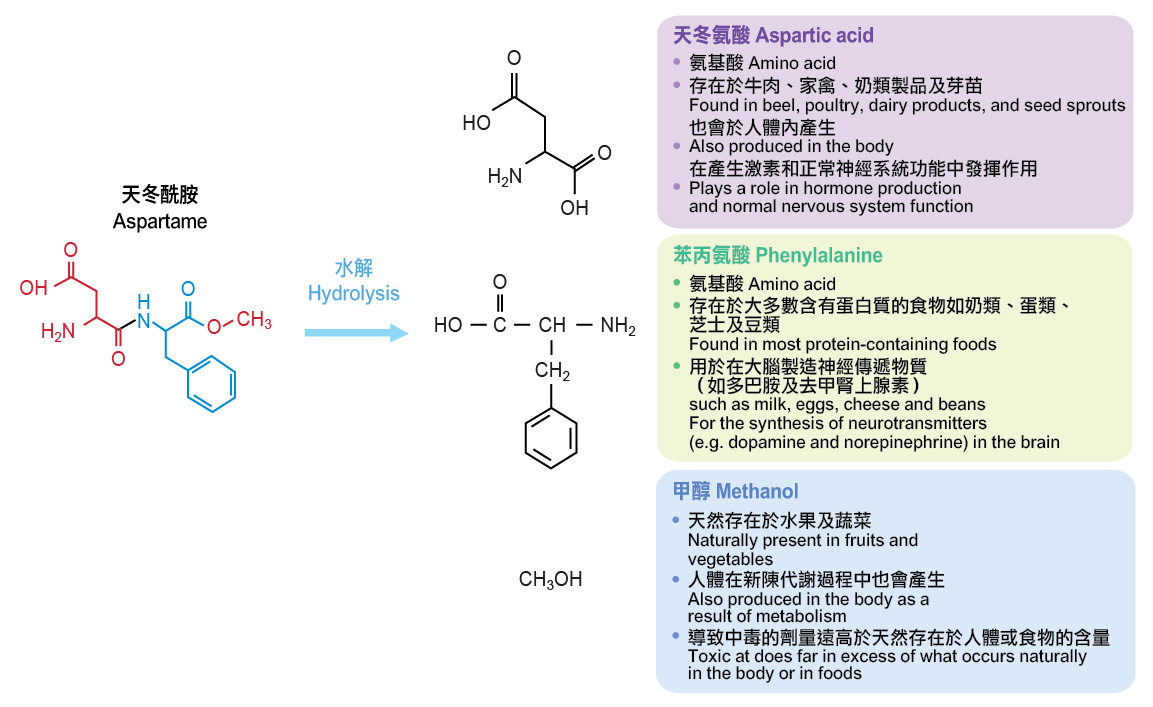
Figure. Hydrolysis of aspartame yields aspartic acid, phenylalanine and methanol
Evaluations of Aspartame by IARC and JECFA
IARC assessed the potential carcinogenic effect of aspartame, while JECFA considered all possible health impacts in its risk assessment, taking dietary exposure of aspartame into consideration. The findings in the joint statement by IARC and JECFA on 14 July 2023 are summarised as follows:
(a) IARC classified aspartame as possibly carcinogenic to humans (Group 2B) on the basis of "limited evidence" for carcinogenicity in experimental animals and humans;
(b) In the three observational studies that allowed an assessment of aspartame and cancer, IARC noted that a positive association was observed between the consumption of artificially sweetened beverages and the risk of liver cancer; however, chance, bias or confounding could not be ruled out as an explanation for the positive findings;
(c) JECFA concluded that there was no convincing evidence from experimental animal or human data that aspartame has adverse effects after ingestion, and it is not possible to establish a link between aspartame exposure in animals and the appearance of cancer. This conclusion is supported by the information that aspartame is fully hydrolysed in the gastrointestinal tract into metabolites that are identical to those absorbed after consumption of common foods; and
(d) JECFA also concluded that dietary exposure to aspartame does not pose a health concern and indicated no reason to change the previously established acceptable daily intake (ADI) of 0–40 mg/kg body weight for aspartame.
The World Health Organization (WHO) will continue to monitor new evidence and encourage independent research groups to develop further studies on the potential association between aspartame exposure and consumer health effects.
How is Aspartame Regulated?
In Hong Kong, sweeteners, including aspartame, approved for food uses have undergone safety evaluations by international authorities such as JECFA, and are regulated under the Sweeteners in Food Regulations (Cap. 132U). Sweeteners should be used under the condition of Good Manufacturing Practice (GMP), that is, the quantity added to food shall be limited to the lowest possible level necessary to accomplish the desired effect.
What Consumers Should Do?
Some consumers use sweeteners as a means for weight control. The key to maintaining body weight is not to consume more calories than those that are burnt. Low-calorie sweeteners can be used as an alternative to reduce calories by replacing table sugar. Nevertheless, they only work if the calories saved are not added back, say, by eating more food later in the day, or by taking in more of other calorie-yielding nutrients such as carbohydrates, protein and fat in the overall diet. Achieving long-term weight maintenance rests on a healthy and active lifestyle that includes a sensible, balanced diet and regular physical activity, and this requires long-term commitment.

