Risk Assessment Studies
Report No. 24
Chemical Hazard Evaluation
DIETARY EXPOSURE TO DDT
OF
SECONDARY SCHOOL STUDENTS
July 2006
Centre for Food Safety
Food and Environmental Hygiene Department
The Government of the Hong Kong Special Administrative Region
Correspondence:
Correspondence:
Risk Assessment Section
Centre for Food Safety
Food and Environmental Hygiene Department
43/F, Queensway Government Offices,
66 Queensway, Hong Kong
Email: enquiries@fehd.gov.hk
Table of Contents
ABSTRACT
This study estimated the dietary exposure to DDT of the secondary school students in Hong Kong and assessed the associated health risks.
Dietary exposure to DDT was estimated using the local food consumption data obtained in secondary school students in 2000 and the concentrations of DDT in food samples taken from the local market. Laboratory analysis for DDT was conducted by the Food Research Laboratory of the Food and Environmental Hygiene Department.
The dietary exposures to DDT for average and high consumers of the secondary school students were 0.145 and 0.291 μg/kg bw/day respectively. Both levels fell well below the Provisional Tolerable Daily Intake (PTDI) of 10 μg/kg bw/day established by the Joint Food Agriculture Organization / World Health Organization Meeting on Pesticide Residues (JMPR). It could be concluded that both the average and high consumers of the secondary school students were unlikely to experience major toxicological effects of DDT.
The results also showed that the food group "seafood", particularly fish and oyster, was the main dietary sources of DDT.
Because of the ubiquitous nature of DDT, low levels of DDT in foods might be unavoidable. Food trade is recommended to observe good agricultural and manufacturing practices to minimise DDT contaminations in food. Members of the public are advised to maintain a balanced diet so as to avoid excessive exposure from a small range of food items, particularly fatty food of animal origin.
OBJECTIVES
This study aims to estimate the dietary exposure to dichlorodiphenyltrichloroethane (DDT) of secondary school students in Hong Kong so as to assess whether there are any risks to their health.
INTRODUCTION
2. The application of chemicals in agriculture and industry gained its popularity since the beginning of the 20th century. Following increasing usage of these chemicals, concerns regarding the possible impacts of chemical contaminants on the environment and human health have been aroused. Among those chemical contaminants, a group of substances called the persistent organic pollutants (POPs) are of particular concern due to their persistence and bioaccumulation in the environment. Both the United Nations Environment Programme 1 (UNEP) and the World Health Organization (WHO) 2 have recommended the monitoring of POPs in human milk for assessing their impact on human health. Local studies have also been carried out in Hong Kong.
3. Of the POPs studied, studies indicated that concentrations of DDT in breast milk samples of Hong Kong mothers gave birth in 1999-2000 were high when compared with those obtained from mothers of other Asian or European countries 3 and ranked the highest among the 26 participating countries/regions in the third WHO Regional Office for Europe coordinated survey of breast milk in 2002-03 4 . There are concerns on the public health implications of dietary exposure to DDT of the population in Hong Kong. Since diet is considered to be the major route of DDT exposure for the general population, the Food and Environmental Hygiene Department (FEHD) undertook to study the dietary exposure to DDT of people in Hong Kong.
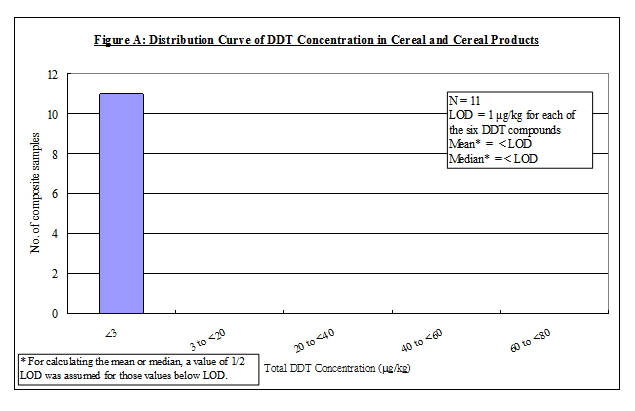
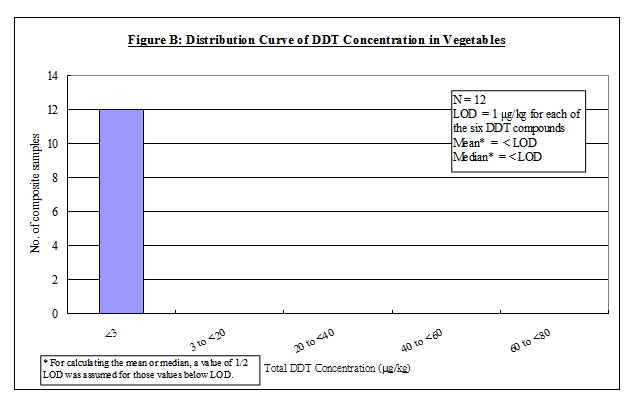
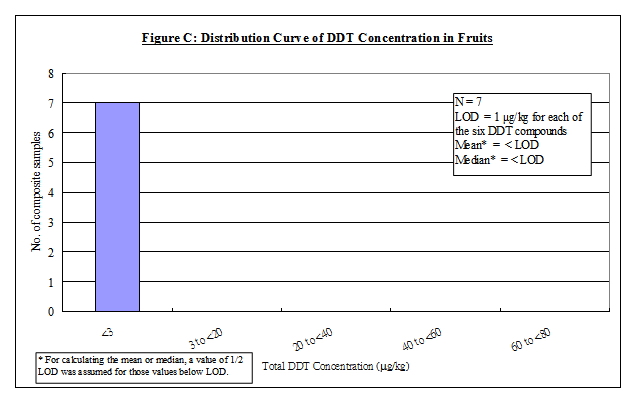
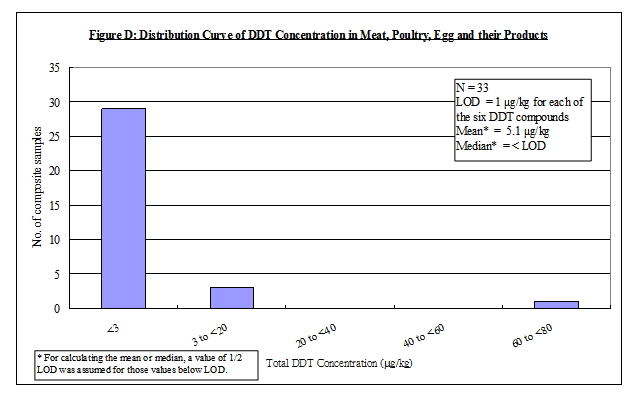
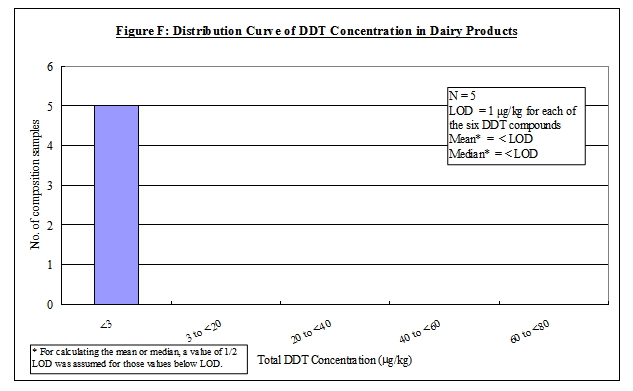
Nature of DDT
4. DDT is an organochlorine insecticide with a broad spectrum of activity 5, 6, 7 . The term DDT usually refers to p,p'-DDT. Commercial DDT pesticides or technical gradea DDT usually contain p,p'-DDT as the major ingredient (65-80%), together with smaller amounts of other compounds, including o,p'-DDT (15-21%), p,p'-dichlorodiphenyldichloroethane (p,p'-DDD) (up to 4%) and the contaminant, 1-(p-chlorophenyl)-2,2,2-trichloroethanol (up to 1.5%). Other forms of DDT, including o,p'-DDD, p,p'- and o,p'- dichlorodiphenyldichloroethene (DDE) are either impurities or metabolites of technical DDT 6, 7. In this report, the term "DDT" would refer to the mixtures of all of the p,p' and o,p' isomers of DDT, DDE and DDD. The chemical identity of the above DDT compounds is listed in Table 1.

5. All DDT compounds are white crystalline solids, tasteless and almost odourless. The odour threshold for DDT in water is 0.35 mg per litre 9.
6. DDT was first synthesised in 1874 but it was not used till its insecticidal properties were discovered in 1939. DDT was used to control insects in forest and on agricultural crops such as the pink boll worm on cotton, codling moth on deciduous fruit, Colorado potato beetle, the European corn borer and so on. Moreover, it was used to control household pests such as fleas, moth and lice, etc. In addition, DDT has been commonly used to control insect-transmitted diseases like malaria, typhus, yellow fever and sleeping sickness 5, 6, 7, 8, 9. Due to its effectiveness, long residual persistence, low acute toxicity and low cost, the application of DDT was once very popular 7.
7. On the other hand, both DDD and DDE are degradation products of DDT. p,p'-DDD was used as an insecticide to an extent much lesser than DDT. o,p'-DDD is used for treating cancer of the adrenal gland whilst DDE has no commercial use 7. DDE has no application.
8. DDT may enter the environment via its production, transport, application and disposal. On the other hand, another technical gradea insecticide, dicofol, have been showed to contain p,p'-DDT and p,p'-DDE as contaminants. Once released, DDT may be present in air, water and soil. DDT can be degraded via atmospheric photooxidation in air, photolysis on the surface of water or soil or biodegraded to form DDE, DDD and other metabolites. DDT and its metabolites can then be adsorbed onto surface soil or particulates in water and become sediments eventually. DDT can also undergo long-range transport through the atmosphere via a process called "global distillation" in which DDT migrates from warmer regions to colder regions through repeated cycles of volatilisation and deposition from/onto soil and water surfaces. This leads to global redistribution of DDT in the environmental media and the presence of DDT metabolites in places where the use of DDT has been greatly restricted. As much as 50% DDT can remain in soil after 10-15 years of application of DDT 6, 7, 10 .
9. Due to their high persistence and lipohilicity, DDT and its metabolites tend to accumulate in fatty tissues of living organisms and their concentration would magnify along the food chain. Hence organisms at higher trophic levels usually contain more DDT than those at lower trophic levels 6, 7, 10 .
10. The use of DDT has been banned in many countries since 1970s because of its persistence and accumulation in the environment after its extensive application. Moreover, its ability of travelling long distances in the air and water as well as its possible adverse effects on the environment and human health attracted global attention to its elimination. DDT was later designated as a POP by the Governing Council of the UNEP in 1997 and given restricted use status under the Stockholm Convention in 2001 6, 7 . Under the Stockholm Convention, the production and use of DDT shall be eliminated unless it is used for disease vector control in accordance with WHO recommendations and guidelines. Those parties to the Convention who would like to apply DDT in their territories should also notify the Secretariat 11 .
Toxicity of DDT
Kinetics and metabolism
11. After oral exposure, DDT is slowly absorbed from the gastrointestinal tract. The presence of fat in food will promote the absorption of low doses of DDT. Some DDT will then be converted to the much less toxic form DDE, which does not undergo further transformation apparently. DDT and its major metabolite, DDE, can be stored in all tissues but tend to accumulate in adipose tissue because of their high lipophilicity. However, the uptake of DDT by fat is slow. Most p,p'-DDE found in human adipose tissue indicates previous dietary exposure of DDT. It was also found that the level of DDT in adipose tissue would reach equilibrium when the rate of intake was constant 6, 7, 8 .
12. Detoxification of DDT in human body involves its conversion into DDD and then to a water soluble product for urinary excretion, which is the main route of excretion of DDT. The whole elimination process is slow at a rate of about 1% of stored DDT per day. DDT can also be eliminated via biliary excretion in the faeces, excretion of human milk or placental transfer 6, 8 .
Acute effects
13. The target organ for acute effects is the nervous system. Experimental animal studies showed that the oral median lethal dose (LD50) for DDT in water suspension or powder ranged from 300 to 2500 mg/kg whilst those for DDT in oil solution ranged from 100 to 800 mg/kg. The presence of fats and oils promotes absorption and enhances the toxicity of DDT subsequently 8 .
14. In humans, onset of signs and symptoms depends on the dose. It has been reported that a single oral dose of 10 mg/kg body weight (bw) DDT produced illness in some individuals and that of 16 mg/kg or greater led to convulsions. It was also reported that the lethal dose in an adult was about 30 g. The earliest symptom of poisoning is hyperaesthesia of the mouth and lower part of the face, followed by paraesthesia of the same area and of the tongue and then by dizziness, an objective disturbance of equilibrium, paraesthesia, tremor of the extremities, confusion, malaise, headache, fatigue and delayed vomiting 8 .
15. The Joint Food Agriculture Organization / World Health Organization Meeting on Pesticide Residues (JMPR) in 2000 considered that DDT is no longer used in agricultural practice but may be present in food commodities as a contaminant because of its persistence in the environment. As peaks of acute dietary intake above the Provisional Tolerable Daily Intake (PTDI) are not likely to occur, an acute reference dose was not allocated .
Genotoxicity and carcinogenicity
16. DDT is not genotoxic 8 . The International Agency for Research on Cancer (IARC) of WHO has evaluated the carcinogenicity of DDT and its associated compounds. The IARC considered that there was sufficient evidence in laboratory animals but inadequate evidence in humans for their carcinogenicity and classified them as Group 2B (possibly carcinogenic to humans) agents 13 . However, JMPR in 2000 reviewed epidemiological data on the association between exposure to DDT and cancer risk and concluded that an increased risk for pancreatic cancer could not be excluded in humans for prolonged occupational exposure to technical-grade DDT 12.
Reproductive or teratogenic effects
17. There was also some evidence of impaired sexual development in male pups in experimental animals after perinatal exposure to p,p'-DDE. The US Agency of Toxic Substances and Disease Registry considered that the DDT complex could impair reproduction and/or development in a number of animal species. Perturbations of reproductive function which were probably due to the activation of oestrogen receptors and inhibition of androgen receptors by DDT-related compounds were observed. Experimental studies suggested that p,p'-DDE is anti-androgenic and that o,p'-DDT is estrogenic. However, the few available data on the reproductive effects did not support the correlation between DDT exposure and endocrine disruption as well as stillbirth, miscarriage or premature rupture of foetal membranes in humans 7, 9, 12 .
Other chronic effects
18. Long term exposure to DDT has been demonstrated to cause liver damage in rats 7, 9, 12 .
Safe reference intake
19. The JMPR in 2000 established a PTDI of 0.01 mg/kg bw for DDT on the basis of the no-observed-adverse-effect level (NOAEL) of 1mg/kg bw per day for developmental toxicity in rats and a safety factor of 100 9, 12 . PTDI is an estimate of the amount of a contaminant that can be ingested over a lifetime without appreciable risk. An intake above the PTDI does not automatically mean that health is at risk. Transient excursion above the PTDI would have no health consequences provided that the average intake over long period is not exceeded as the emphasis of PTDI is a lifetime exposure.
Sources of Human Exposure
20. DDT and its metabolites are ubiquitous in the environment. It has been recognised that dietary intake is the main route of exposure of the general population. It has also been estimated that over 90% of the DDT stored in the general population is derived from food. Fatty foods of animal origin (meat, fish and dairy products), are the major sources of intake of DDT and related compounds 9.
21. On the other hand, exposures to DDT via inhalation or dermal contact have been considered to be negligible due to its low concentration in the atmosphere. Exposure via drinking water is also considered negligible because of the low solubility of DDT in water 7.
SCOPE OF STUDY
22. To estimate the dietary exposure to DDT, this study covered six major food groups, namely (i) cereals and cereal products, (ii) vegetables, (iii) fruits, (iv) meat, poultry, egg and their products, (v) seafood, as well as (vi) dairy products. The selection is based on the occurrence of DDT in these food groups and the consumption patterns.
METHODOLOGY
Food Consumption Data
23. The food consumption data in this report was extracted from the Food Consumption Survey conducted in local secondary school students in 2000 by the FEHD. In the survey, a stratified three-stage sampling plan was used, with a sampling frame of 472 secondary schools and more than 380,000 students, covering almost all the local secondary schools. A total of 967 students from 27 schools participated in the survey yielding a response rate of 77% at the school level and 96% at the student level. The mean weight of the participated students was 52.0 kg 14 .
Sampling Plan
24. Food samples were taken from the local market according to the above six food groups. Food items were selected so as to match those in the Food Consumption Survey as well as those with likely occurrence of DDT. Three samples of each food item were taken randomly from different sources for laboratory analysis.
Laboratory Analysis
25. Laboratory analysis was conducted by the Food Research Laboratory of the Centre for Food Safety. All food samples were treated and analysed as consumed so as to give a better estimate of DDT concentration to be consumed. The three samples collected were mixed and homogenised to give a composite sample. The composite sample was then extracted by cyclohexane, followed by purification using gel permeation chromatography and determination of DDT using gas chromatography/mass spectrometry (GC/MS). The limit of detection (LOD) for each of the six DDT compounds was 1 μg/kg (ppb). The adopted LODs are comparable to those used by authorities in the international arena for dietary exposure assessment and are in fact lower than the ones adopted in the Australian and New Zealand total diet studies.
26. The level of DDT in each composite sample was obtained by the summation of the respective levels of each of the six DDT compounds. When the analytical value was below the LOD, the true value of DDT in the food sample could be anywhere between zero and the LOD. The treatment for these results was particularly important when a large percentage of the analytical results of a particular food group fell below the LOD. While it may not be appropriate to assume a zero concentration for all samples with analytical values below LOD, assigning these non-detects a value of LOD, would, however, grossly overestimate the dietary exposure. In this study, a value of 1/2-LOD was then assigned to all results below LOD. Since the levels of contaminants in food, including DDT, usually follow a log-normal distribution, the approach of assigning a value of 1/2-LOD to all non-detected levels is considered conservative, particularly for food groups in which majority of food items have analytical values below the LOD.
Dietary Exposure
27. Daily dietary exposure from an individual food item was obtained by combining the consumption data and the concentration of an individual food item. Total exposure for each secondary school student was obtained by summing exposures from all food items. The mean and 95th percentile of the daily exposure levels were used to represent the dietary exposure for average and high consumers respectively.
28. The estimated exposure levels were then compared by the PTDI established by the JMPR.
RESULTS
Food Consumption Data
29. Food consumption data for the six food groups are given in Table 2.
Table 2: Food Consumption Patterns for Secondary School Students
| Food groups |
Mean consumption
(g/day) |
|---|---|
| Cereal and cereal products |
478.0
|
| Vegetables |
295.3
|
| Fruits |
309.1
|
| Meat, poultry, egg and their products |
203.7
|
| Seafood |
122.4
|
| Dairy products |
143.2
|
Concentration of DDT in Food
30. A total of 294 food samples were taken and combined into 98 composite samples for analysis. The results are summarised in Table 3.
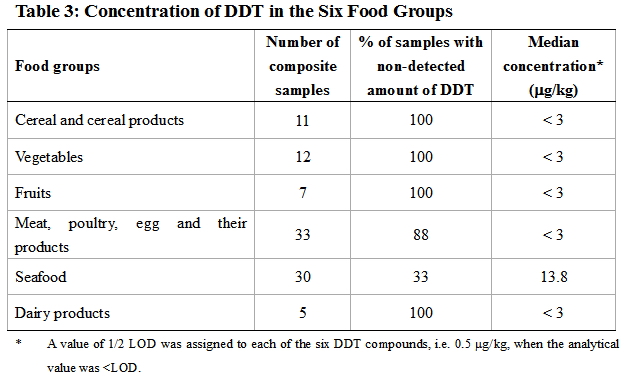
31. DDT was only detected in two food groups, namely "meat, poultry, egg and their products" and "seafood". Distribution curves of DDT concentrations in different food groups are presented in the Annex.
Dietary Exposure
Average secondary school students
32. The dietary exposure to DDT for an average secondary school student was estimated to be 0.145 μg/kg bw/day. The main dietary source of DDT was seafood which contributed to 39% of the total exposure. Dietary exposures to DDT from different food groups are showed in Table 4.
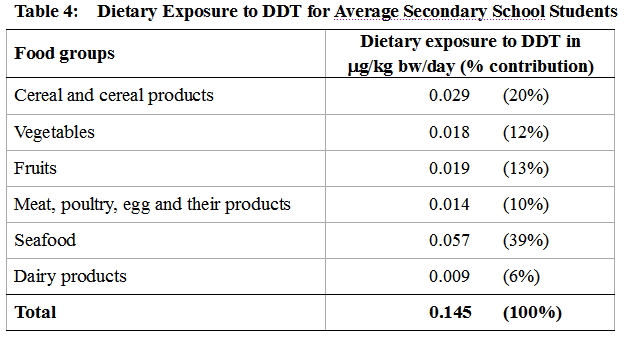
High consumers
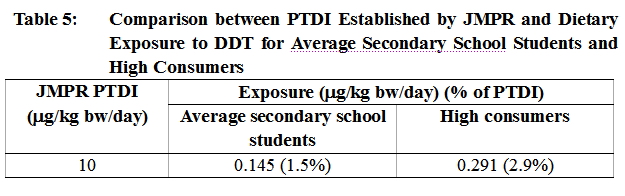
33. Further analysis was undertaken to estimate the risk that high consumers might be exposed to. The 95th percentile exposure level of the secondary school students was used to represent the dietary exposure to DDT for a high consumer and was estimated to be 0.291 µg/kg bw/day (Table 5).
DISCUSSION
Dietary Exposure
34. The dietary exposure to DDT for an average secondary school student was estimated to be 0.145μg/kg bw/day whilst that for the high consumer was 0.291 μg/kg bw/day. These exposures amounted to 1.5% and 2.9% of the PTDI respectively. Exposures to DDT for both an average secondary school student and the high consumer fell well below the PTDI established by the JMPR.
35. The dietary exposure estimates therefore suggested the average secondary school students and high consumers were unlikely to experience major toxicological effects of DDT.
Effects of non-detected values
36. In this study, a value of 1/2-LOD was assigned to all analytical values below LOD. However, the true value of DDT in the food sample could be anywhere between zero and the LOD. To address this issue of uncertainty, DDT concentration in each food sample was also estimated using an upper bound and lower bound estimates. The upper bound was calculated by setting analytical values below LOD to the LOD while the lower bound was calculated by setting analytical values below LOD to zero.
37. Using these upper and lower bound estimates, the dietary exposure to DDT was calculated. The DDT exposure of an average secondary school student would be anywhere between 0.053 (lower bound estimate) and 0.238 (upper bound estimate) μg/kg bw/day while that of high consumers could be anywhere between 0.128 (lower bound estimate) and 0.475 (upper bound estimate) μg/kg bw/day. These exposures amount to 0.5 – 2.4% of PTDI for an average student and 1.3 – 4.8% for the high consumer respectively.
Major dietary sources of DDT
38. In this study, the main dietary source of DDT was "seafood" which contributed to 39% of the total exposure. Fish and oyster are particularly significant contributors to DDT exposures. This pattern matched the results of the total diet studies conducted in the Mainland that aquatic foods (33%) were the main contributor to the total dietary exposure to DDT 15 .
39. Though fish did not have the highest level of DDT (median level: 22.3 μg/kg), its relatively high consumption pattern made it the largest contributor to overall dietary exposure of DDT, i.e. 18%. It was noted that different fish species contained different levels and patterns of DDT compounds.
40. Oysters were found having the highest level of DDT (269.5 μg/kg). Exposure to DDT from oysters accounted for 14% of total dietary exposure and it was one of the key contributors among the food group "seafood".
41. Although DDT residue was not detected in any "cereals and cereal products", its relatively high consumption and the conservative approach being adopted in this study (i.e. to assign a value of 1/2-LOD to non-detected values) made this food group one of the key contributors to the DDT exposure.
Distribution of DDT in food
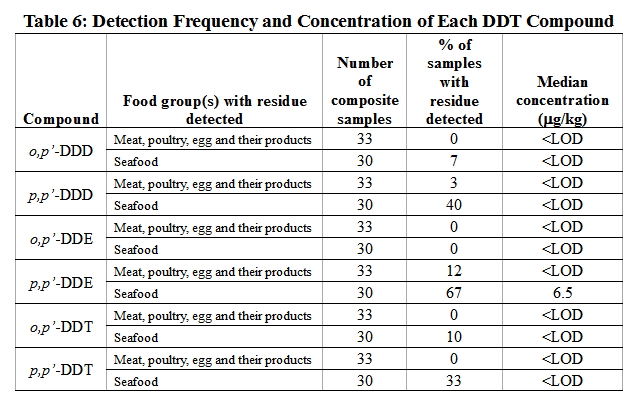
42. p,p'-DDE, the major metabolite of p,p'-DDT, was the most frequently noted residue in detected food samples as most DDT in the environment has been degraded to the stable DDE form whereas DDD has usually been transformed to other compounds. This is consistent with the results of recent overseas studies 16, 17 . On the other hand, low level of p,p'-DDT was detected in some seafood samples even though the use of DDT has been banned in many countries including the Mainland. It might be due to (i) the persistence of DDT; (ii) the long-range transboundary movement of DDT from places where DDT has been used; and (iii) the use of dicofol, which is a permitted pesticide in countries including the Mainland and have been showed to contain p,p'-DDT and p,p'-DDE as contaminants (Table 6).
Effects of food processing
43. Literature indicated that baking, frying, broiling, smoking and microwaving of fish and meat products all effectively reduced the total DDT concentration whereas home-canning reduced p,p'-DDT in tomato paste. It has also been demonstrated that p,p'-DDT, but not p,p'-DDE or p,p'-DDD would decompose upon heating 7 . Since all food samples were treated and analysed as consumed in this study, the effect of cooking has already been taken into account.
International comparison
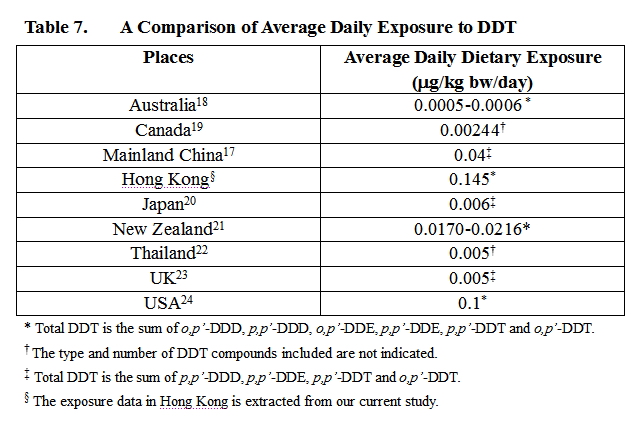
44. Studies on dietary exposure to DDT conducted during 1990-2000s in overseas were reviewed and summarised in Table 7.
45. It can be seen that dietary exposure to DDT of secondary school students in Hong Kong is relatively high when compared with those results from countries such as Australia 18 , Canada 19 , Japan 20 , New Zealand 21 , Thailand 22 , the United Kingdom 23 and the USA 24 . This might explain other research findings 3, 4 that levels of DDT in human milk of Hong Kong mothers are higher than levels in other countries. However, dietary exposures to DDT of both average secondary school students and high consumers fell well below the PTDI established by the JMPR and hence they were not likely to experience major toxicological effects of DDT.
46. Nevertheless, it should be noted that direct comparison of data between different studies has to be done with caution due to the differences in time when the studies were carried out, research methodology, food group categorisation, methods of collection of consumption data, methods of DDT analysis, methods of treating results below the detection limits and the number of DDT compounds being included.
Other Sources of DDT Exposure
47. Apart from food, air and water may also contribute to DDT exposure. A preliminary investigation of persistent organic pollutants in ambient air in Hong Kong by the Environmental Protection Department found that the total amount of DDT and its metabolites ranged from below detection limits to 0.154 ng/m3 during the winter of 2000/2001 25 . On the other hand, the Water Supplies Department reported that the level of DDT in drinking water in Hong Kong for the period April 2004 to March 2005 fell below 0.5 μg/L, which is well below the WHO guideline (1993) of 2 μg/L 26 . Assuming that the respiratory volume of a 60-kg adult is 20 m3/day and the volume of water consumed is 2 L/day respectively, exposure to DDT from air was estimated to be below 0.051 ng/kg bw/day and that from water was estimated to be below 0.017 μg/kg bw/day. This echoed the literature that the main source of DDT exposure was food. Taking exposures from food and water into account, oral exposure to DDT was still far below the PTDI.
Control Measures
48. Environmental control is the primary measure to minimise total exposure to DDT. This involves global efforts in developing regulatory and other mechanisms to ensure that DDT use is restricted to disease vector control; implementing suitable alternative products, methods and strategies to replace the use of DDT; and strengthening health care provision to reduce the incidence of the vector-borne diseases, with the goal of reducing and ultimately eliminating the use of DDT.
49. The Stockholm Convention on Persistent Organic Pollutants, which is an international convention aiming at restricting and ultimately eliminating the production, use, release and storage of POPs including DDT, has been adopted by the international community in 2001. This Convention applies to China, including the Hong Kong Special Administrative Region (HKSAR) with effect from 2004.
50. Under the Stockholm Convention, the production and the use of DDT should be eliminated unless it is used for disease vector control in accordance with WHO recommendations and guidelines. Those parties to the Convention who would like to apply DDT in their territories should notify the Secretariat the amounts and conditions used, as well as follow the recommendations listed under the Convention with the goal of reducing and ultimately eliminating the use of DDT. In consultation with WHO, the continued need for DDT for disease vector control would also be evaluated on the basis of available scientific , technical, environmental and economic information periodically 11 .
51. In Hong Kong, according to the Pesticides Ordinance, Cap. 133, DDT was deregistered on 31 December 1987 and it is no longer a permitted pesticide. The use of DDT has also already been banned in the Mainland.
52. Regarding the aspect of food safety, DDT has already been included in the routine food surveillance programme of the Centre for Food Safety. The Codex Alimentarius Commission has also established extraneous maximum residue limits (EMRLs) for DDT in foods. To monitor the exposure of the population to POPs, including DDT, total diet studies to be conducted at periodic intervals is considered useful.
LIMITATION
53. The methodology for collecting food consumption data may influence the accuracy of the estimates on dietary exposure. In the Food Consumption Survey, the food consumption pattern of secondary school students was collected using a food frequency questionnaire. Although the questionnaire was very comprehensive, it was not possible to cover every single food item, some of which might be relevant to DDT exposure. Furthermore, only the data of consumption pattern for secondary school students is available.
54. Three samples for each food item were combined into one composite sample for laboratory analysis. Although nearly 300 samples were taken in this study, increasing the number of sample for each food item for laboratory analysis could provide a more precise estimate of the average DDT concentration for a particular food item. However, the number of samples taken have to be balanced with the required resources and number of food items to be included.
REFERENCES
- United Nation Environment Programme Chemicals. Guidance for a global monitoring programme on persistent organic pollutants (1st edition). Geneva: UNEP; 2004.
- World Health Organization. Biomonitoring of human milk for persistent organic pollutants (POPs). [cited 5 Jan 2006] Available from: URL: http://www.who.int/foodsafety/chem/pops/en/index.html
- Wong CKC, Leung KM, Poon BHT. and Wong MH. Organochlorine hydrocarbons in human breast milk collected in Hong Kong and Guangzhou. Archives of Environmental Contamination Toxicology 2002; 43: 364-372.
- Wong MH, Leung AOW, Chan JKY and Choi MPK. A review on the usage of POP pesticides in China, with emphasis on DDT loadings in human milk. Chemosphere 2005; 60: 740-752.
- World Health Organization / Food and Agriculture Organization. Data Sheets on Pesticides No. 21 – DDT. WHO/FAO; December 1976. [cited 6 Jan 2006] Available from: URL: http://www.inchem.org/documents/pds/pds/pest21_e.htm
- World Health Organization Regional Office for Europe. Joint WHO/Convention Task Force on the Health Aspects of Air Pollution – health risks of persistent organic pollutants from long-range transboundary air pollution. Geneva: WHO; 2003.
- The U.S. Agency for Toxic Substances and Disease Registry. Toxicological profile for DDT, DDE and DDD. Atlanta; Department of Health and Human Services; September 2002. [cited 10 Aug 2005] Available from: URL: http://www.atsdr.cdc.gov/toxprofiles/tp35.html
- International Programme on Chemical Safety. DDT: Poison Information Monograph 127. Geneva: WHO; 1999. [cited 20 Sep 2004] Available from: URL: http://www.inchem.org/documents/pims/chemical/pim127.htm
- World Health Organization. DDT and its derivatives in drinking-water – background document for development of WHO Guidelines for Drinking-water Quality. Geneva: WHO; 2004.
- International Programme on Chemical Safety. A review of selected persistent organic pollutants. IPCS; December 1995.
- Stockholm Convention on Persistent Organic Pollutants (POPs). Convention text. Available from: URL: http://www.pops.int/default.htm
- JMPR. Pesticide residues in food 2000: DDT. Available from: URL: http://www.inchem.org/documents/jmpr/jmpmono/v00pr03.hm
- International Agency for Research on Cancer. DDT and associated compounds: Vol. 53. Lyon: IARC; 1991. [cited 1 Apr 2002] Available from:
URL: http://www-cie.iarc.fr/htdocs/monographs/vol53/04-ddt.htm - FEHD. Food Consumption Survey 2000. Hong Kong: FEHD; 2001.
- Zhao, YF et. al. Study on of dietary pesticide residues in Chinese residents. Chinese Journal of Epidemiology 2003; 24(3):661-4. [Article in Chinese]
- Cressey, PJ and Vannoort, RW. Pesticide content of infant formulae and weaning foods available in New Zealand. Food Additives and Contaminants 2003; 20(1): 57-64.
- Zhong W, Xu D, Chai Z and Mao X. 2001 survey of organochlorine pesticides in retail milk from Beijing, P. R. China. Food Additives and Contaminants 2003; 20(3): 254-258.
- Food Standards Australia New Zealand. 20th Australian Total Diet Studies. Canberra: FSANZ; 2003.
- Health Canada. Average dietary intakes (ng/kg bw/day) of pesticide residues for Canadians in different age-sex groups from the 1993 to 1996 total diet study. [cited 4 Jul 2005] Available from: URL: http://www.hc-sc.gc.ca/fn-an/surveill/total-diet/intake-apport/pesticide_intake-apport_pesticide_93-96_e.html
- Maitani, T. Evaluation of exposure to chemical substances through foods – exposure to pesticides, heavy metals, dioxins, acrylamide and food additives in Japan. Journal of Health Science 2004; 50(3): 205-209.
- New Zealand Food Safety Authority. 2003/04 New Zealand total diet study – agricultural residues, selected contaminants and nutrients. Wellington: NZFSA; December 2005.
- Vongbuddhapitak A et al. Dietary exposure of Thais to pesticides during 1989-1996. Journal of AOAC International 2002: 85(1): 134-140.
- UK Committee on Toxicity of Chemicals in Food, Consumer Products and the Environment. Risk assessment of mixtures of pesticides and similar substances. London: UK FSA; September 2002.
- Dougherty, CP et al. Dietary exposures to food contaminants across the United States. Environmental Research Section A 2000; 84: 170-185.
- Louie PKK and Sin DW. A preliminary investigation of persistent organic pollutants in ambient air in Hong Kong. Chemosphere 2003; 52:1397-1403.
- Water Supplies Department. Drinking water quality for the period May 2004 - March 2005. Hong Kong: WSD; 2005. [cited 16 Sep 2005] Available from: URL: http://www.wsd.gov.hk/en/html/pdf/wq/drinking_b-e.pdf
Annex