Risk Assessment Studies
Report No. 34
Chemical Hazard Evaluation
Polybrominated Diphenyl Ethers in Foods of Animal Origin
April 2009
Centre for Food Safety
Food and Environmental Hygiene Department
The Government of the Hong Kong Special Administrative Region
Correspondence:
Risk Assessment Section
Centre for Food Safety,
Food and Environmental Hygiene Department,
43/F, Queensway Government Offices,
66 Queensway, Hong Kong.
Email: enquiries@fehd.gov.hk
Table of Contents
| Executive Summary |
| Objectives |
| Background |
| Scope of Study |
| Methods |
| Results |
| Discussion |
| Limitations |
| Conclusion and Recommendation |
| References |
| Annex |
Risk Assessment Studies
Report No.34
Polybrominated Diphenyl Ethers in Foods of Animal Origin
EXECUTIVE SUMMARY
The Centre for Food Safety (CFS) has conducted a study on polybrominated diphenyl ethers (PBDEs) in foods of animal origin aiming to measure the levels of PBDEs in food of animal origin, to estimate the potential dietary exposure to PBDEs of secondary school students in Hong Kong from these foods and to assess the associated health risks.
PBDEs are a group of persistent and bioaccumulative chemicals used as flame retardants. They are extensively used in plastics, polyurethane foam, and textiles. They are anthropogenic (derived from human activities) chemicals and the three main commercial products are Pentabromodiphenyl Oxide or Ether (PentaBDE), Octabromodiphenyl Oxide or Ether (OctaBDE) and Decabromodiphenyl Oxide or Ether (DecaBDE). PBDEs are ubiquitous in the environment and are potentially toxic. Animal studies found that PBDEs affect brain and reproductive organs, neurobehavioural development, thyroxine level and are toxic to liver. The main sources of PBDE exposure for humans include indoor air, house dust, and food, including human milk. It was estimated house dust accounted for 82% of the overall estimated intakes of PBDEs in the USA. PBDEs have been found rapidly increase in concentration in blood, tissue and breast milk of humans. They have received increasing attention because of their potential to impact upon human health and the environment.
PBDE contamination can be caused by e-wastes and production of brominated fire retardants. Studies have shown the PBDE concentrations in seafood in Hong Kong and aquatic environments in Pearl River Delta were higher than those reported in other countries. In the absence of local data on dietary exposure to PBDEs, the associated health risks to the local population are not clear.
The Study
Sampling was carried out from May to August 2008 from local market. A total of 24 PBDE congeners were analysed in 100 composite samples from 300 individual food items including (i) fish, (ii) seafood and seafood products (other than fish), (iii) meat and meat products, (iv) poultries, (v) eggs, and (vi) dairy products. Laboratory analysis was conducted by the Government Laboratory.
Daily dietary exposures to PBDEs from an individual food item were obtained by combining the consumption data of secondary school students and the levels of PBDEs of that food item. The mean and the 95th percentile of the daily exposure levels were used to represent the dietary exposure for average and high consumer respectively.
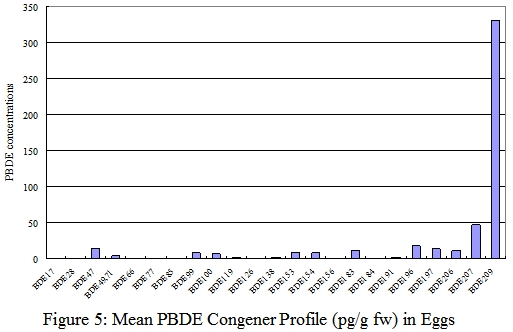
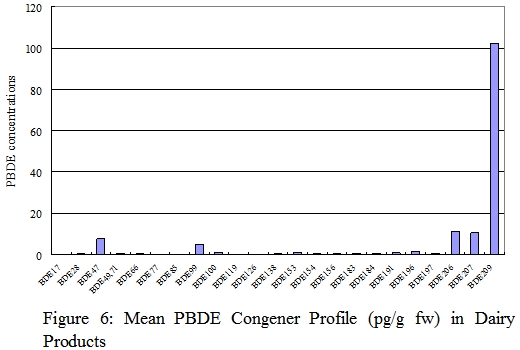
Results
In our study, PBDEs were detected in all samples of food of animal origins tested. The dominant congeners found were BDE-47 and BDE-209. The total PBDE levels varied widely among items from the same food group. The PBDE levels on fresh weight basis of fish ranged from 13-6,600 pg/g, of seafood and seafood products (other than fish) ranged from 15-1,200 pg/g, of meat and meat products ranged from 23-3,500 pg/g, of poultries ranged from 68-670 pg/g, of eggs ranged from 280-800 pg/g, and of dairy products ranged from 12-480 pg/g.
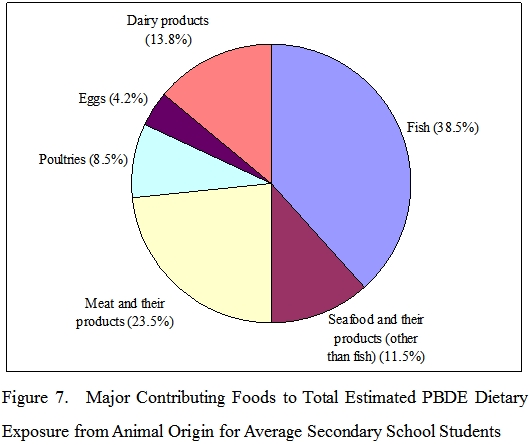
Highest PBDE levels on a fresh weight basis were found in fish. Fish was the major dietary source of PBDEs of secondary school students and contributed to 38.5% PBDE exposure among food from animal origin.
The dietary exposures to PBDEs from food of animal origin for the average and high consumers of secondary school students were estimated to be 2.6 and 6.4 ng/kg bw/day, respectively. According to the JECFA, for the more toxic PBDE congeners, adverse effects would be unlikely to occur in laboratory animals at doses of less than approximately 100 μg/kg bw per day. The margin of exposure (MOE) was used to assess the health risk of dietary PBDE exposure in this study. The MOE is calculated by dividing the dose at which adverse effects were observed in laboratory animal studies by the estimated exposure to PBDEs from food. The lower the MOE, the greater is the public health risk. The calculated MOE values (38,000 for average consumers and 16,000 for high consumers) showed that the estimate exposures of secondary school students were far below any adverse effect dose observed in laboratory animals.
Conclusion and recommendation
On the basis of the available data and taking into account all the inherent uncertainties and limitations it can be concluded that the health risk of secondary school students arising from dietary exposure to PBDEs in food from animal origin is unlikely to be of public health and food safety significance.
Advice to consumers
The present data on PBDEs in foods from animal origin are not sufficient to warrant changes in basic dietary advice on healthy eating which is to maintain a balanced diet so as to avoid excessive exposure to chemical contaminants from a small range of food items. In addition, PBDEs tend to accumulate in the fat of animals. To reduce dietary exposure to PBDEs, the public is advised to consume low-fat products, to trim fat from meat and meat products, to reduce the amount of animal fat used in food preparation and to use cooking methods that reduce fat.
Advice to the trade
Trade members should observe good agricultural and manufacturing practices such as to avoid exposing animals to PBDE sources i.e. foams, insulations, and other fire-retardant materials, to obtain food supplies from reliable sources, and to maintain proper records to enable source tracing when required.
Risk Assessment Studies –
Polybrominated Diphenyl Ethers in Foods of Animal Origin
OBJECTIVES
This study aims to examine the levels of polybrominated diphenyl ethers (PBDEs) in food of animal origin, to estimate the potential dietary exposure to PBDEs of secondary school students in Hong Kong from these foods and to assess the associated health risks.
BACKGROUND
2. PBDEs are a group of anthropogenic chemicals consisting of 209 structurally related chemicals, or congeners that are added to a wide variety of consumer/commercial products (e.g. plastics, polyurethane foam, textiles) in order to improve their fire resistance. 1 These chemicals have received increasing attention to date because of their potential to impact upon human health and the environment.
3. PBDEs have been produced since the 1960s, primarily as three main commercial products: Pentabromodiphenyl Oxide or Ether (PentaBDE), Octabromodiphenyl Oxide or Ether (OctaBDE) and Decabromodiphenyl Oxide or Ether (DecaBDE). PentaBDE is mainly used in polyester and flexible polyurethane foam formulations in amounts that can result in the finished material being composed of up to 30% by weight of flame retardant. OctaBDE is mainly used in thermoplastic resins, in particular ABS (acrylonitrile-butadiene-styrene) plastic, which can contain up to 12% by weight OctaBDE. DecaBDE is used in various plastic polymers such as polyvinyl chloride, polycarbonates and high-impact polystyrene, as well as back coating for textiles (commercial furniture, automobile fabrics, carpets, etc.). 1
4. PBDEs are ubiquitous contaminants with potential for persistence in the environment and strong tendency towards bioaccumulation. PentaBDE and OctaBDE have met the basic criterion of persistent organic pollutants (POPs) and are currently being reviewed under the Stockholm Convention on Persistent Organic Pollutants.2,3
5. Release of PBDEs into the environment can occur during production, application and / or disposal. PBDEs are lipophilic and are being stored in the body fat of animals. They can bioaccumulate along the food chain. Available information indicated that the main sources of PBDE exposure for humans include indoor air, house dust and food, including human milk. 1 A recent review of studies relating to the US population found that unlike other POPs, the key route of human exposure of PBDEs is house dust rather than food. House dust accounted for 82% of the overall estimated intakes of PBDEs.4 Overseas studies found that the levels of total PBDEs in human samples (blood, tissue and breast milk) have increased more than 100 times in the past 30 years with a doubling time of about 5 years.5 On the other hand, recent studies in Sweden have demonstrated a reduction in levels of PBDEs in breast milk. This is thought to be a result of a decline in the use of products containing PBDEs in Sweden. 6,7
6. To address the food safety concern of PBDEs, food safety authorities have conducted toxicological evaluation and dietary survey of PBDEs in food. The Joint Food and Agriculture Organization / World Health Organization Expert Committee on Food Additives (JECFA) evaluated the safety of PBDEs in 2005.1 National food authorities such as Australia have also undertaken studies of PBDE levels in food. 8
7. Locally, according to the Environmental Protection Department (EPD), the higher levels of PBDEs were found in Deep Bay and Victoria Harbour , which may be attributed to effluent discharge, urban runoff and local pollution. DecaBDE was the dominant congener form, accounting for 80-90% of the total PBDEs. The concentrations of the more toxic Penta- and OctaBDEs in sediments were low, ranging 0.1-1.2 μg/kg dry weight.9
8. PBDE contamination can be caused by e-wastes and production of brominated fire retardants. A study revealed that the levels of PBDEs of riverine runoff samples from the Pearl River Delta were in the high end of the global PBDE concentrations in the aquatic environments. 10 Besides, studies showed that mussel samples from the coastal water of Hong Kong and fish from local market contained comparatively high levels of PBDEs. 11,12 In the absence of local dietary exposure data on PBDEs, the associated health risk to the local population is not clear. A study on PBDEs in food of animal origins is needed to examine the situation in the local scene.
Toxicity and Health Effects
9. The JECFA 1, the National Industrial Chemicals Notification and Assessment Scheme (NICNAS) of the Australian Government,13 the European Commission14-16 and the United States Environmental Protection Agency (U.S. EPA) 17-22 have evaluated the toxicity of PBDEs. The toxicity of PBDEs is briefly summarized below:
Kinetics and metabolism
10. Animal studies showed that individual PBDE congeners in the tetrabrominated to hexabrominated range are readily absorbed orally and partition to breast milk. The absorption of PBDEs is directly related to the extent of bromination of the parent diphenly ether; as a general rule, greater substitution with bromination leads to a decrease in bioavailability. 1
11. The metabolism of PBDEs consists of hydroxylation and methoxylation reaction and, in the case of congeners with a higher degree of bromination, oxidation debromination. The excretion in rats is mainly faecal, although adult mice have been shown to readily excrete BDE-47 (tetrabrominated) in urine. The difference in excretion routes results in different half lives for this congener in rodents. Limited pharmacokinetic data were available for humans. On the basis of the observed increase in concentrations of PBDEs in tissue with time, PBDEs are absorbed and bioaccumulated. By extrapolation of the results in rats, the human half life has been estimated to be in the range of several years for individual tetrabrominated to hexabrominated congeners.1,13,14
Acute toxicity
12. The acute toxicity of mixtures of PBDEs is low in rodents. There are no observable effects in standard tests for acute toxicity after exposure to DecaBDE and OctaBDE at doses of several grams per kilogram of body weight, although certain effects (increased mortality, behavioural symptoms and changes in gross pathology) are seen after exposure to PentaBDE at similar high dose.1 The oral median lethal dose (LD 50) of PentaBDE ranged from 2,640 to 6,200 mg/kg body weight (bw) .1,13
Chronic effects
13. PentaBDE and OctaBDE were found to cause liver enlargement in experimental animals, which maybe connected to increased activity of microsomal enzyme in the liver. A number of studies found that PBDEs affect the development, particularly on the brain and the reproductive organs in experimental animals. Changes in neurobehavioural development and thyroxine (thyroid hormone) level were also observed.1,13, 14
14. No epidemiological studies of PBDEs effects in humans have been reported. Little information is available on the overall effects of PBDEs in humans. Several studies have examined the relationship between thyroid hormone status in humans and the serum PBDE levels, but no association was established. 1,13, 14
Genotoxicity
15. The results of the majority of tests for genotoxicity performed in vitro and limited data from test in vivo indicated that PBDE mixtures and individual congeners are not genotoxic. 1,14, 15,16
Carcinogenicity
16. The International Agency for Research on Cancer (IARC) of the World Health Organization in 1999 considered DecaBDE not classifiable as to its carcinogenicity to humans (Group 3) but it has not evaluated the carcinogenicity of PentaBDE and OctaBDE. 23 The NICNAS of Australia considered that there was insufficient information on the carcinogenic potential of commercial PentaBDE and OctaBDE.13 The U.S. EPA in 1990 considered that there was limited evidence for DecaBDE causing liver tumours in animals and there was no human data for evaluation. It then classified DecaBDE as a possible human carcinogen.22
Safety reference value
JECFA
17. The JECFA in 2005 considered that the available data on PBDEs were not adequate for allocating a safety reference value. Nevertheless, the JECFA stated from limited toxicity data that for the more toxic [less brominated] PBDE congeners, adverse effects would be unlikely to occur in rodents at dose of less than approximately 100 μg/kg bw/day. 1
U.S. EPA
18. On the other hand, the U.S. EPA established a chronic oral reference dose of 0.002 mg/kg bw/day for PentaBDE, based on a no-observed-adverse-effect-level (NOAEL) of 1.77 mg/kg bw/day for induction of hepatic enzymes in experimental animals and an uncertainty factor of 1,000. 17 The US EPA also established a chronic oral reference dose of 0.003 mg/kg bw/day for OctaBDE, based on a NOAEL of 2.51 mg/kg bw/day for induction of hepatic enzymes in experimental animals and an uncertainty factor of 1,000. 18
19. As for individual congener, the U.S. EPA established reference dose for chronic exposure (RfD) of 0.0001 mg/kg bw/day for BDE-47 and BDE-99 respectively, based on a benchmark dose lower confidence limit (BMDL) for neurobehavioural effects19,20 ; 0.0002 mg/kg bw/day for BDE-153 and 0.007 mg/kg bw/day for BDE-209 based on a no-observed-adverse-effect-level (NOAEL) for neurobehavioral effects. 21,22
Regulatory Control of PBDEs
20. At present there is no local or international regulatory limit for PBDEs in food. As for the production and use of PBDE-containing products, some developed countries have taken action to control the exposure. For example, the European Union has banned the use of PentaBDE and OctaBDE in all products since August 200424 and has phased out the use of DecaBDE in electronics by July 2006.25 The Australian Government has adopted a precautionary approach by prohibiting the import and/or manufacture of the OctaBDE (except for use as a laboratory standard for analytical determination) and PentaBDE in the country since 2007. 26 In the USA, the EPA reached a voluntary agreement with the sole U.S. manufacturer of PentaBDE and OctaBDE under which that company ceased production of these chemicals by the end of 2004. 27 In Hong Kong, there is no specific regulatory control on PBDEs in the environment under the purview of EPD. Nevertheless, PBDEs were added to the list of toxic substances monitored in marine sediment in 2005 by EPD. In addition, unwanted PBDEs are classified as chemical waste which is subject to control under the Waste Disposal Ordinance.
SCOPE OF STUDY
21. To estimate the dietary exposures to PBDEs from food of animal origin, this study covered six major food groups, namely (i) fish, (ii) seafood and seafood products (other than fish), (iii) meat and meat products, (iv) poultries, (v) eggs, and (vi) dairy products. The selection was based on the reported occurrence of PBDEs in these food groups and the local consumption patterns.
METHODS
Sampling
22. Sampling was carried out from May to August 2008. A total of 300 food samples were taken from the local market according to the above six food groups. Food items were selected based on the results of the Food Consumption Survey conducted in local secondary school students in 2000 by the FEHD28 and the likely occurrence of PBDEs. Three samples of each food item taken randomly from different sources were combined to one composite sample for laboratory analysis.
Laboratory analysis
23. Laboratory analysis was conducted by the Government Laboratory. All samples were analysed as purchased. The edible portion was homogenized or mixed well. A sufficient amount of sample (from 1 g to 150 g depending on the matrix of the food sample concerned) was then weighed. Sodium sulfate and stable isotope (carbon-13 labelled) internal standard of nine PBDE congeners were spiked quantitatively into this measured amount of sample. A dichloromethane: hexane (1:1) mixture was added to the sample and this sample mixture was homogenized again. The resulting slurry was passed through an acidified silica gel column in which dehydration, defatting and extraction was carried out.
24. After solvent evaporation, the concentrated sample extract was cleaned up by small silica column before subjecting to carbon column fractionation. The fraction containing the PBDE congeners was then concentrated by vacuum evaporation and further clean-up by a silica column and an alumina column. The eluate was then dried down by nitrogen evaporation. The residue was reconstituted in 20 μL of carbon-13 injection standard in nonane.
25. This 20 μL final extract was then analyzed by Gas Chromatography / High-resolution Mass Spectrometry (GC/HRMS). The analytes were separated by GC and detected by HRMS set at a resolution of ≥ 5000 in Selected Ion Requisition (SIR) mode. Identification was confirmed by comparing the retention time, masses and ion-abundance ratio of two exact m/z for each of the analytes with those of the corresponding compounds in the calibration standards.
26. The analysis included 24 individual congeners (including tri-, tetra-, penta-, hexa-, hepta-, octa-, nona-, and deca-brominated diphenyl ethers) and these are summarized in Table 1. These congeners were chosen as they are commonly found in food and the environment and also had validated methods of analysis. The reporting limits (RLs) for BDE-17, BDE-28, BDE-47, BDE-66, BDE-77, BDE-85, BDE-99, BDE-100, BDE-119 and BDE-126 were 0.25 pg/g; for BDE-49/71, BDE-138, BDE-153, BDE-154, BDE-156, BDE-183, BDE-184, BDE-191, BDE-196 and BDE-197 were 0.50 pg/g; for BDE-206, BDE-207 and BDE-209 were 1.25 pg/g; and the RL for fat content was 0.5% w/w. The samples were analysed on a fresh weight (whole weight) basis and concentrations reported in picograms per gram (ppt).
27. The concentrations of PBDE congeners 28, 47, 99, 153, 154, 183, 197, 207 & 209 were determined using the isotopic dilution technique. Those for the other PBDE congeners were determined using the internal standard technique. The concentrations of PBDE congeners 49 and 71 were reported as a sum of the two congeners, due to incomplete chromatographic separation of the two peaks under the specific running conditions. The total PBDE concentration was calculated by summing all the concentrations of the 24 individual congeners tested.
Table 1: Individual PBDE Congeners Analysed
 |
|
Congener number |
Bromine substitution pattern |
 |
|
|
BDE-17 |
2,2',4-tribromodipenyl ether |
|
BDE-28 |
2,4,4'-tribromodipenyl ether |
 |
|
|
BDE-47 |
2,2',4,4'-tetrabromodipenyl ether |
|
BDE-49 |
2,2',4,5'-tetrabromodipenyl ether |
|
BDE-66 |
2,3',4,4'-tetrabromodipenyl ether |
| BDE-71 | 2,3',4',6-tetrabromodiphenyl ether |
| BDE-77 | 3,3',4,4'-tetrabromodipenyl ether |
 |
|
|
BDE-85 |
2,2',3,4,4'-pentabromodiphenyl ether |
|
BDE-99 |
2,2',4,4',5-pentabromodiphenyl ether |
|
BDE-100 |
2,2',4,4',6-pentabromodiphenyl ether |
|
BDE-119 |
2,3',4,4',6-pentabromodiphenyl ether |
|
BDE-126 |
3,3',4,4',5-pentabromodiphenyl ether |
 |
|
|
BDE-138 |
2,2',3,4,4',5'-hexabromodiphenyl ether |
|
BDE-153 |
2,2',4,4',5,5'-hexabromodiphenyl ether |
|
BDE-154 |
2,2',4,4',5,6'-hexabromodiphenyl ether |
|
BDE-156 |
2,3,3',4,4',5-hexabromodiphenyl ether |
 |
|
|
BDE-183 |
2,2',3,4,4',5',6-heptabromodiphenyl ether |
|
BDE-184 |
2,2',3,4,4',6,6'-heptabromodiphenyl ether |
|
BDE-191 |
2,3,3',4,4',5',6-heptabromodiphenyl ether |
 |
|
|
BDE-196 |
2,2',3,3',4,4',5,6-octabromodiphenyl ether |
|
BDE-197 |
2,2',3,3',4,4',6,6'-octabromodiphenyl ether |
 |
|
|
BDE-206 |
2,2',3,3',4,4',5,5',6-nonabromodiphenyl ether |
|
BDE-207 |
2,2',3,3',4,4',5,6,6'-nonabromodiphenyl ether |
 |
|
|
BDE-209 |
2,2',3,3',4,4',5,5',6,6'-decabromodiphenyl ether |
 |
|
Food consumption data
28. The food consumption data in this report was extracted from the Food Consumption Survey. In the survey, a stratified three-stage sampling plan was used, with a sampling frame of 472 secondary schools and more than 380,000 students, covering almost all the local secondary schools. A total of 967 students from 27 schools participated in the survey yielding a response rate 77% at the school level and 96% at the student level. The mean body weight of the participated students was 52.0 kg.
Estimation of dietary exposure
29. Daily dietary exposures to PBDEs from an individual food item were obtained by combining the consumption data and the levels of PBDEs of that food item. Total exposure for each secondary school students was obtained by summing exposures from all food items. The mean and the 95th percentile of the daily exposure levels were used to represent the dietary exposure for average and high consumer respectively.
30. The estimated exposure levels of secondary school students to PBDE from foods of animal origin were then compared with the value (100 μg/kg bw per day) used by JECFA for calculating the respective MOEs.
RESULTS
Concentrations of PBDEs in food
31. A total of 100 food items were tested for PBDEs. The test results for each food group are summarized in Table 2 whereas those for each composite food samples are presented in Annex.
Table 2: Concentrations of Total PBDEs in Different Food groups on pg/g Fresh Weight Basis
| No. of samples | Mean | Range | |
|---|---|---|---|
| Fish | 44 | 920 | 13-6,600 |
| Seafood and seafood products (other than fish) | 17 | 190 | 15-1,200 |
| Meat and meat products | 26 | 330 | 23-3,500 |
| Poultries | 5 | 260 | 68-670 |
| Eggs | 3 | 490 | 280-800 |
| Dairy products | 5 | 140 | 12-480 |
| Total | 100 |
Note: Results are rounded to 2 significant figures.
32. According to the WHO's recommendation on evaluation of low-level contamination of food, 29 half of LOD is used for all results less than LOD when less than or equal to 60% of results are below LOD. In our study, 42% of test results for PBDE congeners were "not detected" (below reporting limit, RL). The concentrations of PBDE congeners for each food sample and hence the dietary exposure estimates for total PBDEs were calculated by setting analytical values below RL to RL/2.
33. PBDEs were detected in all food groups. The total PBDE levels varied widely between items from the same food group. Even for the same species, a large variation of PBDE levels existed. It was found that the composite sample of snakehead fish had PBDE level up 6,700 pg/g in fresh weight basis where a re-test of three other individual samples had an average PBDE level at 300 pg/g only. This variation might due to different degrees of contamination in feed or environment among different snakehead samples. The PBDE level (3,400 pg/g) of snakehead fish shown in Annex is the average level of the composite and individual samples.
34. The average and maximum of total PBDE concentrations in fresh weight basis of fish were significantly higher than those of other food groups. The levels on fresh weight basis ranged from 13-6,600 pg/g of which Black cod (6,600 pg/g), Mackerel (3,600 pg/g), Snakehead (3,400 pg/g), Mangrove snapper (2,900 pg/g), Salmon (2,300 pg/g), and Cardinal fish (2,200 pg/g) were relatively high in PBDEs. Since the PBDE concentration of fish is affected by various factors, it is not able to conclude whether marine or freshwater fish will contain higher level of PBDEs.
35. PBDEs are fat-soluble. A higher body fat content would lead to more PBDEs accumulate in the organism. However, there was only a weak linear relationship (r=0.46) between the PBDE levels and fat contents of the different types of fish tested in this study. This indicated that other factors such as living environment, species variation, diet, age, and metabolism may also influence the PBDE level in fish.
36. For food samples other than fish, only BBQ pork was relatively high in PBDEs (3,500 pg/g). Since the rest of the pork products including those with high fat content in this study were not high in PBDEs, the high level found in BBQ pork could due to contamination of feed and environment of individual animal. Also, contamination of the pork by PBDE-containing sources during processing cannot be ruled out.
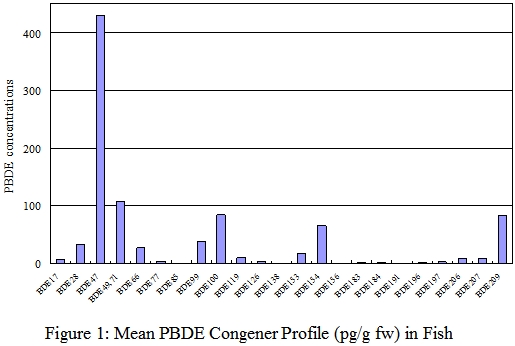
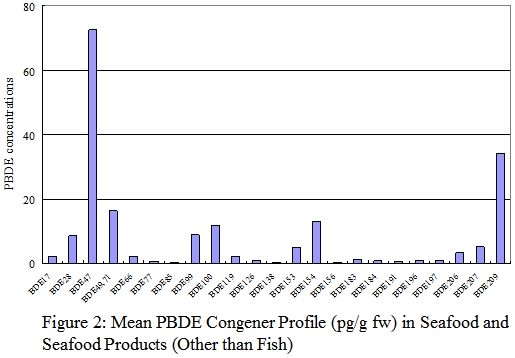
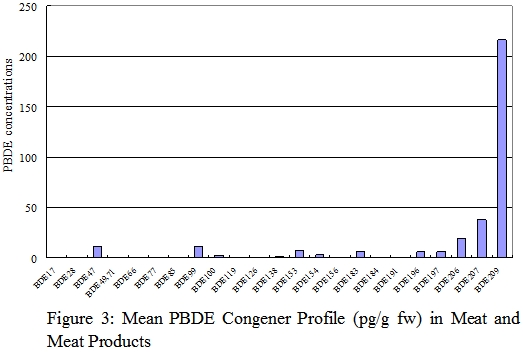
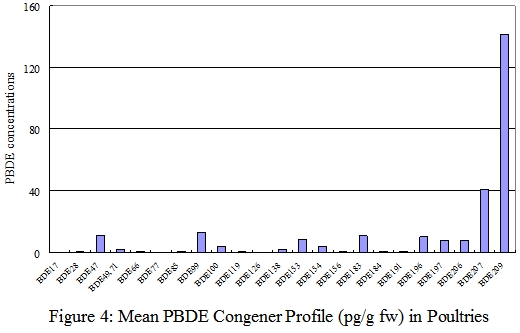
PBDE Congener profiles
37. The PBDE congener profiles for each food groups for fresh weight analysis were shown in figures 1-6. Details of the PBDE congener profiles for individual food samples can be found in Annex. The most commonly reported dominant congeners in organisms were BDE-47, -99, -100, -153, and -154.1 On the other hand, some recently published literatures revealed that BDE-209, the major congener in commercial PBDE production that are currently used in plastic polymers and textiles was the dominant congeners in certain foods such as dairy and meat products. 8,30
38. In this study, BDE-47 (tetrabrominated) and BDE-209 (decabrominated) appeared to be the predominant congeners in food available in Hong Kong . The most abundant congener in most fish, seafood and seafood products was BDE-47. These two food groups were also relatively high in BDE-100, BDE-154, and BDE-209. For meat and meat products, poultries, egg and their products, and dairy products, BDE-209 was the predominant congener. One of the explanations to the difference between the PBDE profiles of aquatic and terrestrial organisms is that the bioavailability of highly brominated congeners BDE-209 may be low in aquatic organisms due to its large molecular size. 31
39. The PBDE congener profiles of fish, meat and dairy products in our study were similar to those reported in the Australia and USA. 8,30 The PBDE congener profiles of individual samples of meat, poultry, egg, and dairy products were consistence with their respective group profiles. However, the profiles of some fish and seafood samples were different from their group profiles, with BDE-209 instead of BDE-47 as the predominant congener. This may due to the variation in species and living environment of the fish and seafood samples.
Comparison of PBDE concentrations in foods from other countries
40. Table 3 shows the PBDE concentrations in selected foods in Hong Kong compared with those from other countries. The PBDE levels of foods available in Hong Kong were in line with those reported in other countries. However, one should be cautious when making comparison between these results since there are differences in foods sampled, analytical methodologies used and number of congeners tested as well as the assumptions made in calculation and reporting of the data.
41. A local study conducted by Cheung KC et al. found that the levels of PBDEs in market fish samples collected in 2004 ranged from 0.53 to 130 ng/g (530 to 130,000 pg/g) wet (fresh) wt. 12 Although the PBDE levels of some fish samples we found were comparable to those reported in the study, the overall level of PBDEs in fish samples in our study were lower, ranged from 13 to 6,600 pg/g. This difference would be due to variations in time of sampling, and sample such as species, source, and size. A recent review study suggested that factors like different sampling times or tissues chosen for analyses can severely influence the PBDE content found in the fish. 32
Table 3: Comparison of PBDE Mean Values for Selected Food Items in pg/g Fresh (Wet) Weight with Other Countries
| Food | Mean concentration of PBDEs (pg/g) | |||
|---|---|---|---|---|
|
Hong Kong ND=1/2 RL |
Australia 8 ND=1/2 LOQ |
USA 30 ND=1/2LOD |
Spain 33 ND=1/2LOQ |
|
| Fresh Fish | 920 | 190.3 | 1120 | 553.5 |
| Beef (1) | 56 | 193.0 | 147.3 | 42 |
| Pork (2) | 126 | 699.4 | 131 | 172 |
| Lamb (3) | 500 | 366.1 | 186 | 31 |
| Chicken (4) | 108 | 287.1 | 206 | 10 |
| Sausage (5) | 220 | 358.5 | 918.2 | N/A |
| Hen egg | 280 | 932.1 | 85 | 64 |
| Milk, whole | 16 | 36.9 | 7.9 | 24 |
| Ice cream | 480 | 118.9 | 171 | N/A |
| Yoghurt (6) | 160 | 89.7 | 31.6 | N/A |
Notes:
|
- |
1
|
=
|
Beef in HK included beef, ox belly and beef steak; Australia sample was beef steak; USA sample included ground beef and beef tenderloin; Spain sample included beef and beef products.
|
|
- |
2
|
=
|
Pork in HK included pork chop, lean pork, pork belly, pork neck meat, sparerib; Australia sample was pork chops, grilled, USA sample included pork and ground pork; Spain sample was pork and pork products.
|
|
- |
3
|
=
|
Lamb in HK was lamb slices, Australia sample was lamb chop, loin, grilled; in USA sample was ground lamb; Spain sample was lamb.
|
|
- |
4
|
=
|
Chicken in HK included chicken breast, chicken wing, chicken drumstick; Australia sample was chicken breast; USA sample included chicken breast and ground chicken, Spain sample was chicken;
|
|
- |
5
|
=
|
Sausages in HK included big red sausage and sausage, Australia sample was beef sausage, USA sample included pork sausage, sausage, and wieners.
|
|
- |
6
|
=
|
Yoghurt in USA was low fat.
|
|
- |
RL
|
=
|
reporting limit
|
|
- |
LOD
|
=
|
Limit of detection
|
|
- |
LOQ
|
=
|
Limit of quantification
|
|
- |
Results of HK study are rounded to 2 significant figures.
|
||
Food consumption data
42. Food consumption data for the six food groups are given in Table 4.
Table 4: Food Consumption Pattern for Secondary School Students
| Food groups | Mean consumption (g/day) |
|---|---|
| Fish | 53.6 |
| Seafood and seafood products (other than fish) | 70.9 |
| Meat and meat products | 100.4 |
| Poultries | 80.4 |
| Eggs | 14.6 |
| Dairy products | 142.2 |
Dietary exposure to PBDEs
43. The dietary exposure to PBDEs from food of animal origin for the average consumer of secondary school student was estimated to be 2.6 ng/kg bw/day. The main dietary source of PBDEs was "fish" followed by "meat and meat products" which contributed to 38.5% and 23.5% of the total exposure from food of animal origin, respectively. Dietary exposures to PBDEs from different food groups are shown in Figure 7.
44. Further analysis was undertaken to estimate the risks that higher consumers might be exposed to. The 95th percentile exposure level of the secondary school students was used to represent the dietary exposure to PBDEs for a high consumer and was estimated to be 6.4 ng/kg bw/day.
45. The current estimates of dietary PBDE exposure by JECFA was approximately 4 ng/kg bw/day.1 Recent studies showed that the PBDE exposure from dietary sources for adults in the Australia was 1-132 ng/kg bw/day8, in the USA was 0.9-1.2 ng/kg bw/day30, in Spain was 1.2-1.4 ng/kg bw/day33 On the other hand, Cheung KC et al, estimated the PBDE exposure through fish alone by people in Hong Kong was 311-1677 ng/day12 which is equivalent to 5.2-28.0 ng/kg bw/day, assuming an average adult weight of 60 kg. Our estimated PBDE exposures from food from animal origins (2.6-6.4 ng/kg bw/day) were comparable to those reported by overseas countries but lower than that of Cheung KC et al. The PBDE exposures estimated by other local and overseas' studies are shown in table 5.
Table 5: Comparison of PBDE Exposures Estimated by Other Local and Overseas' Studies.
|
|
PBDE exposures from dietary sources |
|
HK ( CFS) |
2.6- 6.4 a |
|
Australia 8
|
upper bound 49-132 (1) |
|
USA 30 |
0.9-1.2 |
|
Spain 33 |
1.2-1.4 |
|
HK (Cheung KC et al.) |
5.2-28 b, (4) |
Note:
|
- |
a
|
:
|
Exposures from food of animal origin only.
|
|
- |
b
|
:
|
Exposures from fish only.
|
|
- |
(1)
|
:
|
The LOQ assigned to all results below the LOQ (non-detections)
|
|
- |
(2)
|
:
|
50% LOQ value assigned to all results below the LOQ.
|
|
- |
(3)
|
:
|
Zero value assigned to all results below the LOQ.
|
|
- |
(4)
|
:
|
The estimated daily PBDE exposure through fish by people in Hong Kong by Cheung KC et al. was 311-1677 per/day. The exposure was estimated assuming an average adult weight of 60 kg.
|
46. In the absence of sufficient data to establish a tolerable weekly or monthly intake for PBDEs, the margin of exposure (MOE) was used to determine the health risk of dietary PBDE exposure of secondary school students from food of animal origin. The MOE is calculated by dividing the dose at which adverse effects were observed in laboratory animal studies by the estimated exposure to PBDEs from food. The lower the MOE, the greater is the public health risk. According to the JECFA, the limited toxicity data suggested that for the more toxic PBDE congeners, adverse effects would be unlikely to occur in rodents at doses of less than approximately 100 μg/kg bw per day. This value (100 μg/kg bw/day) has been used as the basis for determining the magnitude of the MOE for all PBDEs. The dietary exposure to PBDEs from food of animal origin for an average consumer of secondary school students was estimated to be 2.6 ng/kg bw/day whilst that for the high consumer was 6.4 ng/kg bw/day. The resulting MOE for an average consumer was 38000 (100 [ μg/kg bw/day] x 1000/2.6 [ng/kg bw/day]) and for high consumers was 16000 (100 [ μg/kg bw/day] x 1000 /6.4 [ng/kg bw/day]). Such MOE values suggested that the estimated exposure to PBDEs from food of animal origin for both average and high consumers of secondary school students were of low concern for human health.
Table 6: MOEs for Dietary Exposures to PBDEs for Average and High Consumers of Secondary School Students
|
Dose that would unlikely to cause adverse effect in rodents as estimated by JECFA |
Average consumer
|
High consumer
|
|---|---|---|
|
<100 |
38,000 |
16,000 |
Note: Results are rounded to two significant figures.
DISCUSSION
47. In our study, PBDEs were detected in all samples of food of animal origins tested. The total PBDE levels varied widely between items from the same food group. Beside fat content, other factors such as species variation, habitat, diet, age, and metabolism might also influence the PBDE level in the organism. The PBDE levels found and estimated dietary exposures of PBDEs were similar to those reported from other countries.8,30,33 Fish was the highest in PBDE levels on a fresh weight basis and was the major source of PBDEs of the diet of secondary school students.
48. The MOE values for an average and high consumers suggested that there was a large margin (over 10,000-fold) between the dose that would unlikely to cause adverse effect in animals (<100 μg/kg bw/day) and the estimated exposure to PBDEs from food of animal origin of secondary school students (2.6 and 6.4 ng/kg bw/day). Thus dietary exposure to PBDEs in food from animal origin is unlikely to be of public health and food safety significance.
49. A recent study on PBDE concentrations in human samples of South China indicated that the concentrations were within the range reported in Europe. 34 However, PBDEs are persistent and bioaccumulative flame retardants. If their production and application are not reduced, it is expected that the concentration of PBDEs in food and human will be increased. Therefore, continuously monitoring of the PBDE levels in food is important to public health of local people.
LIMITATIONS
50. Although 100 composite samples were taken in this study, increasing the number of sample for each food item for laboratory analysis could provide a more precise estimate of the average concentrations of PBDEs for a particular food item. However, the number of samples taken have to be balanced with the required resources and number of food items to be included. The food items tested did not cover all of PBDE-containing foods, so that the dietary exposure might be underestimated.
51. Limited by available resources, all samples were analysed as purchased in our study. Except for some ready-to-eat meat and dairy products, most food samples were raw. Studies showed that it was unlikely that PBDEs were formed or lost during usual cooking processes. However, changes in PBDE content can be expected to be seen on a fresh weight basis owing to changes in fat and water content. 1 PBDE concentrate in the fat portions of the food, such as the fatty tissue of the fish and cooking might reduce the PBDE levels in food, but there are no data available.
52. The methodology for collecting food consumption data may influence the accuracy of the estimates on dietary exposure. In the Food Consumption Survey, the food consumption pattern of secondary school students was collected using a food frequency questionnaire. Although the questionnaire was very comprehensive, it was not possible to cover every single food item, some of which might be relevant to exposure to PBDEs. For instance, the food frequency questionnaire included consumption data for fish but consumption pattern of individual fish species or other seafood species was not known. Furthermore, only data of consumption pattern for secondary school students is available.
CONCLUSION AND RECOMMEDATIONS
53. On the basis of the available data and taking into account all the inherent uncertainties and limitations , it can be concluded that the health risk of secondary school students arising from dietary exposure to PBDEs in food from animal origin is unlikely to be of public health and food safety significance.
54. The present data on PBDEs in foods from animal origin are not sufficient to warrant changes in basic dietary advice on healthy eating which is to maintain a balanced diet so as to avoid excessive exposure to chemical contaminants from a small range of food items. In addition, like other POPs, PBDEs tend to accumulate in the fat of animals. To reduce dietary exposure to PBDEs , the public is advised to consume low-fat products, to trim fat from meat and meat products, to reduce the amount of animal fat used in food preparation and to use cooking methods that reduce fat.
55. Trade members should observe good agricultural and manufacturing practices such as to avoid exposing animals to PBDE sources ie. foams, insulations, and other fire-retardant materials, to obtain food supplies from reliable sources, and to maintain proper records to enable source tracing when required.
REFERENCES
1JECFA. 64 th Meeting – Safety evaluation of certain contaminants in food. WHO Food Additives Series 55/ FAO Food and Nutrition Paper 82; Geneva , 2006. Available from URL:
http://whqlibdoc.who.int/publications/2006/9241660554_PDE_eng.pdf
2Persistent Organic Pollutants Review Committee of the Stockholm Convention on Persistent Organic Pollutants. Risk profile on commercial pentabromodiphenyl ether –Report of the Persistent Organic Pollutants Review Committee on the work of its second meeting. Geneva : UNEP; 2006. Available from URL:
http://www.pops.int/documents/meetings/poprc_2/meeting_docs/report/POPRC-2%20rep%20add1.pdf
3 Persistent Organic Pollutants Review Committee of the Stockholm Convention on Persistent Organic Pollutants. Decision POPRC-26: Commercial octabromodiphenyl ether. Available from: URL:
http://www.pops.int/documents/meetings/poprc_2/evaluation2/annex_i2_6.pdf
4 Lober M. Exposure of Americans to polybrominated diphenyl ethers. Journal of Exposure Science and Environmental Epidemiology; 18, 2-19. 2008.
5 Hites RA. Polybrominated diphenyl ethers in the environment and in people: A meta-analysis of concentrations. Environmental Science Technology; 38(4), 945-956, 2004.
6 N oren K, Meirontye D. Certain organochorine and organobromin contaminants in Swedish human milk in perspective of past 20-30 years. Chemosphere; 40, 1111-1123. 2000.
7Guvenius DM et al. Human prenatal and postnatal exposure to polybrominated diphenyl ethers, polychlorinated biphenyls, polychlorobiphenyols, and pentachlorophenol. Environmental Health Perspective; 111(.9) , 1235-1241, 2003. Available from URL:
http://www.ehponline.org/members/2003/5946/5946.html
8 Food Standards Australia New Zealand . Polybrominated diphenyl ethers (PBDE) in food in Australia . 2007. Available from URL:
http://www.foodstandards.gov.au/_srcfiles/PBDE_Report_Dec_07.pdf
9 Environmental Protetction Department of the Government of the Hong Kong SAR. Environment in HK: Marine water quality-Water dio_fur. 2008. Available from URL:
http://www.epd.gov.hk/epd/textonly/english/environmentinhk/water/marine_quality/
Dio_fur.html
10 Guan YF et al. Riverine inputs of polybrominated diphenyl ethers from the Pearl River Delta ( China ) to the coastal ocean. Environ mental . Science. Technology; 41(17), 6007-6013, 2007.
11 Ramu K et al. Asian mussel watch program: Contamination status of polybrominated diphenyl ethers and organochlorines in coastal waters of Asian countries. Environ mental . Science Technology; 41(13), 4580-4586. 2007.
12 Cheung KC et al. Exposure to polybrominated diphenyl ethers associated with consumption of marine and freshwater fish in Hong Kong . Chemosphere; 70, 1707-1720, 2008.
13 National Industrial Chemicals Notification and Assessment Scheme (NICNAS). Interim public health risk assessment of certain PBDE congeners. Canberra : Common Wealth of Australia . March 2007. Available from URL:
http://www.nicnas.gov.au/publications/car/Other/Final%20Interim%20Report%20-%20March.pdf
14 European Communities. "European Union Risk Assessment Report: Diphenyl ether, pentabromo derivative (pentabromodiphenyl ether)". 2001. Available from URL:
http://ecb.jrc.it/DOCUMENTS/Existing-Chemicals/RISK_ASSESSMENT/REPORT/
penta_bdpereport015.pdf
15 European Communities. "European Union Risk Assessment Report: Bis(pentabromophenyl) ether". 2002. Available from URL:
http://ecb.jrc.it/DOCUMENTS/Existing-Chemicals/RISK_ASSESSMENT/REPORT/
decabromodiphenyletherreport013.pdf
16 European Communities. "European Union Risk Assessment Report: Diphenyl ether, octabromoderivative". 2003. Available from URL:
http://ecb.jrc.it/DOCUMENTS/Existing-Chemicals/RISK_ASSESSMENT/REPORT/
octareport014.pdf
17 US EPA. Integrated Risk Information System – Pentabromodipheyl ether. [cited 4 Nov 2008 ] Available from URL: http://www.epa.gov/iris/subst/0184.htm
18 US EPA. Integrated Risk Information System – Octabromodipheyl ether. [cited 4 Nov 2008 ] Available from: URL: http://www.epa.gov/iris/subst/0180.htm
19 US EPA. Integrated Risk Information System – 2,2',4,4'-Tetrabromodiphenyl ether (BDE-47). (CASRN 5436-43-1) [cited 4 Nov 2008 ] Available from URL:
http://www.epa.gov/iris/subst/1010.htm
20 US EPA. Integrated Risk Information System – 2,2'4,4',5-Pentabromodiphenyl ether (BDE-99). (CASRN 60348-60-9) [cited 4 Nov 2008 ] Available from URL:
http://www.epa.gov/iris/subst/1008.htm
21 US EPA. Integrated Risk Information System – 2,2'4,4',5,5'-Hexabromodiphenyl ether (BDE-153) (CASRN 68631-49-2) [cited 4 Nov 2008 ] Available from URL:
http://www.epa.gov/iris/subst/1009.htm
22 US EPA. Integrated Risk Information System – 2,2',3,3',4,4',5,5',6,6'-Decabromodiphenyl ether (BDE-209). (CASRN 1163-19-5). [cited 4 Nov 2008 ] Available from URL: http://www.epa.gov/iris/subst/0035.htm
23 IARC. Summaries & Evaluations: Decabromodiphenyl oxide, Volume 81, 1999. Available from URL:
http://www.inchem.org/documents/iarc/vol71/079-decabrdipoxide.html
24 European Commission. Directive 2003/11/EC of the European Parliament and of the Council of 6 February 2003 amending for the 24th time Council Directive 76/769/EEC relating to restrictions on the marketing and use of certain dangerous substances and preparations (pentabromodiphenyl ether, octabromodiphenyl ether). February 2003. Available from URL:
http://eur-lex.europa.eu/LexUriServ/site/en/oj/2003/l_042/l_04220030215en00450046.pdf
25 European Commission. Directive 2002/95/EC the European Parliament and of the Council of 27 January 2003 on the restriction of the use of certain hazardous substances in electrical and electronic equipment. February 2003. Available from URL:
http://eurlex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2003:037:0019:0023:EN:PDF
26 Australian Government Department of Health and Ageing NICNAS. NICNAS Information Sheet PBDEs: Interim public health risk assessment report on certain PBDE Congeners contained in commercial preparations of pentabromodiphenyl ether and octabromodiphenyl ether. March 2007. Available from URL:
http://www.nicnas.gov.au/Publications/CAR/Other/PBDE_PDF.pdf
27 US EPA. Pollution Prevention and Toxics: Polybrominated diphenylethers (PBDEs) significant new use rule (SNUR) questions. August 2007. Available from URL:
http://www.epa.gov/oppt/pbde/pubs/qanda.htm
28 FEHD. Food Consumption Survey 2000. Hong Kong : FEHD; 2001.
29 WHO. GEMS/Food-EURO Second Workshop on reliable evaluation of low-level contamination of food – report of a workshop in the frame of GEMS/Food-EURO. WHO; May 1995. Available from URL:
http://www.who.int/foodsafety/publications/chem/lowlevel_may1995/en/index.html
30 Schecter A et al. Polybrominated diphenyl ether levels in an expanded market survey of U.S. food and estimated PBDE dietary intake by age and sex. Environmental Health Perspectives; 114, 1515-1620, 2006. Available from URL:
http://www.ehponline.org/members/2006/9121/9121.html
31 Eljarrat E et al. Occurrence and bioavailability of polybrominated diphenyl ethers and hexabromocyclododecane in sediment and fish from the Cinca river, a tributary of the Ebro river ( Spain ). Environmental Science & Technology; 38(9), 2603-2608, 2004.
32 Frederisksen M et al. Human internal and external exposure to PBDEs—A review of levels and sources. International Journal of Hygiene and Environmental Health; 212(2), 109-134, 2009
33 Bocio A et al. Polybrominated diphenyl ethers (PBDEs) in foodstuffs: Human exposure through the diet. Journal of Agricultural and Food Chemistry; 51, 3191-3195, 2003.
34 Bi X et al. Polybrominated diphenyl ethers in South China maternal and fetal blood and breast milk. Environmental Pollution; 144, 1024-1030, 2006.
Annex : PBDE Concentrations (pg/g) and Fat Content (% w/w) in Foods on Fresh Weight Basis.