Risk Assessment Studies
Report No. 23
Ochratoxin A in Food
May 2006
Centre for Food Safety
The Government of the Hong Kong Special Administrative Region
Table of Contents
Conclusions and Recommendations
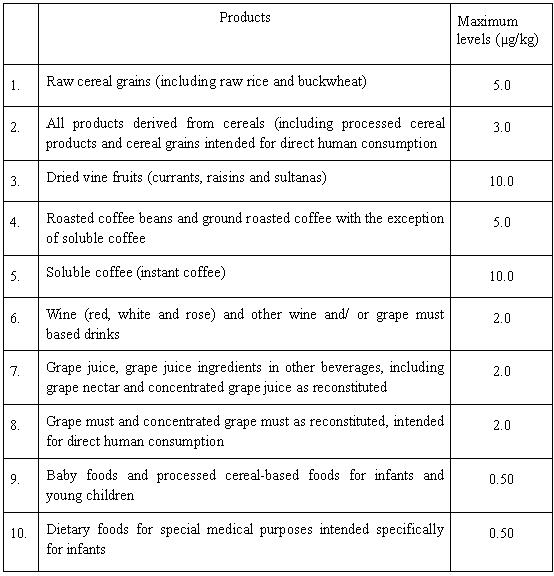
Annex 1 : Legal limits for ochratoxin A in different food products set by the European Commission
Annex 2: Levels of ochratoxin A in various food groups
ABSTRACT
Ochratoxin A is a toxic and potentially carcinogenic fungal toxin found in a variety of food commodities.
This study conducted by the Food and Environmental Hygiene Department aims to assess the levels of ochratoxin A in foods in Hong Kong and the levels of exposure in secondary school students to this substance. A total of 287 food samples were taken from the local retail market for ochratoxin A analysis. Majority of food samples including local staple food of steamed rice, noodles and congee were not found to contain measurable amounts of ochratoxin A. Presence of measurable amount of ochratoxin A was reported in 101 food items (35% of all samples) with levels ranged from 0.01 – 2.09 mg/kg, all of which were below the draft Codex standard of 5 mg/kg.
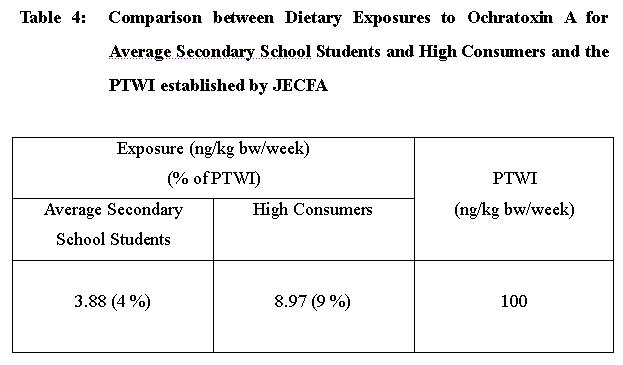
Dietary exposures to ochratoxin A for average and high consumers of secondary school students were estimated to be 3.88 and 8.97 ng/kg body weight/week respectively, both levels were well below the provisional tolerable weekly intake (PTWI) of 100 ng/kg body weight established by JECFA.
The results suggested that both the average and high consumers of secondary school students are unlikely to experience major toxicological effects of ochratoxin A.
OBJECTIVES
This study aims to determine the prevailing levels of ochratoxin A in food available in the local market in Hong Kong, and to investigate the dietary exposure to ochratoxin A in secondary school students.
BACKGROUND
Nature of ochratoxin A
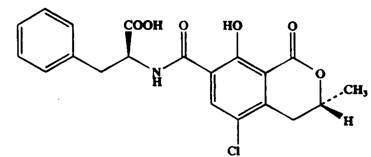
2. Ochratoxins are a group of mycotoxins produced as secondary metabolites by several fungi of the Aspergillus or Penicillium families and are weak organic acids consisting of a derivative of an isocoumarin (Figure 1). The family of ochratoxins consists of three members, A, B, and C which differ slightly from each other in chemical structures. These differences, however, have marked effects on their respective toxic potentials. Ochratoxin A is the most abundant and hence the most commonly detected member but is also the most toxic of the three 1, 2, 3 , It is a potent toxin affecting mainly the kidney. As in other mycotoxins, ochratoxin A can contaminate a wide variety of foods as a result of fungal infection in crops, in the field during growth, at harvest, in storage and in shipment under favourable environmental conditions especially when they are not properly dried. Ochratoxin A may be present in a foodstuff even when the visible mould is not seen.
Figure 1. Ochratoxin A
Occurrence and common food products involved
3. Ochratoxin A is found mainly in cereal and cereal products. This group of commodities has been reported to be the main contributors to ochratoxin A exposure in exposure assessments carried out by the European Commission 4, 5 , accounting for 50% of total dietary exposure of ochratoxin A in European countries (SCOOP task 3.2.7, 2002).
4. Besides cereals and cereal products, ochratoxin A is also found in a range of other food commodities, including coffee, cocoa, wine, beer, pulses, spices, dried fruits, grape juice, pig kidney and other meat and meat products of non-ruminant animals exposed to feedstuffs contaminated with this mycotoxin. Ruminant animals such as cows and sheep are generally resistant to the effects of ochratoxin A due to hydrolysis to the non-toxic metabolites by protozoa in the stomachs before absorption into the blood 6 .
Associated fungal species and geographical distributions
5. The frequency of the occurrence of the different species of ochratoxin A- producing fungi differs according to the geographical regions and in the commodities affected. The Penicillium species that is associated with ochratoxin A production, Penicillium verrucosum, is a common storage fungus and is the source of ochratoxin A in crops in the cool temperate regions such as Canada, eastern and north western Europe and parts of South America. It grows only at temperatures below 30°C and at a lower water activity 7 . Penicillium species may produce ochratoxin at temperatures as low as 5°C 8 .
6. In contrast, Aspergillus species appears to be limited to conditions of high humidity and temperature growing in the tropical and subtropical climates and is the source of contamination for coffee and cocoa beans, spices, dried vine fruit, grape juice and wine. Aspergillus ochraceus is the best known species of ochratoxin –producing Aspergillus. It grows at moderate temperatures and at a high water activity and is a significant source of ochratoxin A in cereals. It infects coffee beans usually during sun-drying causing contamination in green coffee.
7. Aspergillus carbonarius is highly resistant to sunlight and survives sun-drying because of its black spores and therefore grows at high temperatures. It is associated with maturing fruits and is the source of ochratoxin A in grapes, dried vine fruits, and wine and is also another source of ochratoxin A in coffee.
8. Another closely related species, Aspergillus niger, is another minor source of ochratoxin A production in infected coffee beans and dried vine fruits.
Chemistry and effects of processing
9. Ochratoxin A is a moderately stable molecule and is able to survive most food processing to some extent and may thus occur in consumer products. Processing may involve boiling, baking, roasting or fermentation, and the degree to which it is destroyed will further depend on other parameters such as pH, temperature and the other ingredients present. Ochratoxin A is only partly destroyed during cooking and bread making. Baking and roasting have been reported to reduce the toxin content by a mere 20% 9, 10 , However, physical treatment of grain, such as scouring while cleaning the grain prior to milling, can result in a >50% reduction of ochratoxin A contamination in the resultant wheat flour. Milling seems to have no or only a minor effect on the level of ochratoxin A 11 .
Sources of human exposure
10. Dietary intake represents the main source of ochratoxin A in human. Human exposure to ochratoxin A occurs mainly through consumption of contaminated crops or food derived from animals exposed to contaminated feedstuffs. Occupational exposures from inhalation of dust at grain storage warehouses are uncommon. Levels of ochratoxin A in human can be measured by detection of ochratoxin A in human blood and breast milk. A collaborative survey carried out by 13 member states in European Commission in 1995 (SCOOP task 3.2.2) estimated ochratoxin A intakes in human based on plasma levels of the toxin as a biomarker and found that similar levels were obtained by estimations from dietary exposure from food surveys. This suggests that the main sources of ochratoxin A are the known dietary sources covered in the food surveys.
Toxicity and health implications
Metabolism
11. Ochratoxin A is absorbed from the gastrointestinal tract. In most species, ochratoxin A is absorbed from the stomach as a result of its acidic properties 12 . Absorption also takes place in the small intestine particularly in the proximal jejunum. In non-ruminant species such as pigs, chickens, rabbits and rats, around half of the ingested ochratoxin A may be absorbed 13, 14 , The absorbed ochratoxin A is distributed via blood, mainly to the kidneys, and at lower concentrations to the liver, muscle and fat, with a proportion metabolised into the non-toxic metabolite ochratoxin alpha and other less toxic minor metabolites at various sites in different species 12, and a significant proportion excreted unchanged. Ochratoxin A has a long serum half-life in non-ruminant animals and in humans (72-120 h in pigs, 840 h in a human subject) on the basis of its strong binding to serum macromolecules.
12. In ruminant species such as the cow, effective hydrolysis of ochratoxin A to the non-toxic ochratoxin alpha takes place in the four stomachs in the presence of the ruminant protozoa 6 rendering the species resistant to the effects of the toxin.
13. Transfer to the milk has been demonstrated in rats, rabbits and humans. In contrast, little ochratoxin A is transferred to the milk of ruminants, again due to metabolism of this mycotoxin by the rumen microflora.
Acute toxicity
14. The acute toxicity of ochratoxin A is relatively low, although large species differences and sensitivity are seen with oral LD50 values ranging widely in different species. Oral LD50 values has been demonstrated to range from 0.2 mg/kg bw in dogs, 1 mg/kg bw in pigs, 3.3 mg/kg bw in chicken, and 46-58 mg/kg bw in mouse. Dogs and pigs have been reported to be the most sensitive species . Effects of acute poisoning such as multifocal haemorrhages in various organs and fibrin thrombi in the spleen, brain, liver, kidney and heart have been reported following single dose administration. Nephrosis, hepatic and lymphoid necrosis, and enteritis with villous atrophy have also been observed in the test species 16, 17 , At present, there are no documented cases of acute toxicity reported in humans.
Chronic toxicity
15. The subchronic and chronic effects of ochratoxin A are of greatest concern. Ochratoxin A has been shown to be nephrotoxic, hepatotoxic, teratogenic and immunotoxic to several species of animals and carcinogenic in mice and rats causing tumours of the kidney and liver.
Nephrotoxicity
16. In particular, its role in chronic nephropathies has been extensively documented in many mammalian species. Ochratoxin A is considered the causal agent in nephropathies observed in several species of agricultural animals, particularly in pigs. It has produced nephrotoxic effects in all species of single - stomach animals studied so far, even at the lowest level tested (200 μg/kg feed in rats and pigs).
17. Ochratoxicosis in farm animals such as pigs and poultry may lead to pathological changes in the kidney such as tubular atrophy, interstitial fibrosis and hyalinised glomeruli. The main target site of ochratoxin A toxicity is the renal proximal tubule, where it exerts cytotoxic and carcinogenic effects. Significant sex and species differences in sensitivity to nephrotoxicity have been observed where pigs have been found to be a more sensitive species compared with rats or mouse.
18. In human, dietary exposure to ochratoxin A in parts of Bulgaria, Romania and the former Yugoslavia may have association with Balkan endemic nephropathy, a chronic progressive kidney disease, that is characterised by progressive hypercreatinaemia, uraemia, hypertension and oedema. 20
Other toxicities
19. Ochratoxin A has been known to cause hepatic damage and hepatic necrosis in experimental animals. It is a potent teratogen in mice, rats, hamsters and chicken. Both teratogenic and reproductive effects have been demonstrated.
20. Ochratoxin A has been reported to be an immunosuppressor and affects the immune system in a number of mammalian species. It was able to cause inhibition of protein biosynthesis and inhibition of macrophage migration 18 .
Carcinogenicity
21. Ochratoxin A was tested for carcinogenicity by oral administration in mice and rats. Increased incidence of hepatocellular tumours in mice of each sex, and association with renal-cell adenomas and carcinomas have been reported in male mice and in rats of each sex fed with ochratoxin A. In 1993, the International Agency for Research on Cancer (IARC) classified ochratoxin A as possible human carcinogen (Group 2B) and concluded that there was sufficient evidence in experimental animals for the carcinogenicity of ochratoxin A and inadequate evidence in humans for the carcinogenicity of ochratoxin A 19. The doses at which carcinogenicity were observed in rodents had been reported to be higher than those that caused nephrotoxicity.
Genotoxicity
22. Ochratoxin A has been shown to induce DNA damage, DNA repair, and chromosomal aberrations in mammalian cells in vitro as well as DNA damage and chromosomal aberrations in mice treated in vivo. However, the mechanism for genotoxicity is unclear and there was no evidence that it is mediated by direct interaction with DNA.
Observations in humans
23. Human exposure, as demonstrated by the occurrence of ochratoxin A in blood, and in human milk, has been observed in various countries in Europe. Ochratoxin A was found more frequently and high concentrations in blood samples obtained from people living in regions where the fatal human kidney disease, Balkan Endemic Nephropathy, occurs. A highly significant relationship has been observed between Balkan endemic nephropathy and tumours of the urinary tract, particularly with tumours of the renal pelvis and ureters. Nevertheless, similar average concentrations have been found in some other European countries where this disease is not observed. The Joint FAO/WHO Expert Committee on Food Additives (JECFA) concluded in 2001 that the epidemiological and clinical data available do not provide a basis for calculating the likely carcinogenic potency in human and that Balkan Endemic Nephropathy may involve other nephrotoxic agents. 18
Level of safe intake of ochratoxin A
24. Following the evaluations carried out in 1990, 1995 and 2001 for ochratoxin A, JECFA has established a provisional tolerable weekly intake (PTWI) of 100 ng/kg bw/wk for this substance. 20
25. The European Commission's Scientific Committee for Food (SCF), after reviewing its opinion on ochratoxin A, concluded in 1998 that it would be prudent to reduce exposure to ochratoxin A as much as possible, ensuring that exposures are towards the lower end of the range of tolerable daily intakes which has been estimated by other bodies, at a level below 5 ng/kg bw/ day. 21
26. Tolerable intake, which can be expressed in daily, weekly (e.g. PTWI) or monthly basis is an estimate of the amount of a contaminant that can be ingested over a lifetime without appreciable risk. An intake above PTWI does not automatically mean that health is at risk. Transient excursion above the PTWI would have no health consequences provided that the average intake over long period is not exceeded as the emphasis of PTWI is a lifetime exposure.
27. Values for the estimated dietary exposures to ochratoxin A in European countries range from 0.13 to 4.6 ng/kg bw per day (i.e. 0.91 to 32.2 ng/kg bw per week) with the major source being cereal and cereal products (SCOOP task 3.2.2 and SCOOP 3.2.7). Following the evaluation in 2001 by JECFA, the mean total intake of ochratoxin A at the international level was estimated to be 45 ng/kg bw per week based on aggregated data. This was assessed on the basis of data on mean consumption combined with weighted mean level of contamination 18 .
Codex draft limit
28. In the recent sessions of the Codex Committee of Food Additives and Contaminants (CCFAC), there had been active discussions on setting the draft maximum level for ochratoxin A in raw wheat, barley, rye and derived products. A draft maximum level of 5 μg/kg is now held at step 7, pending for JECFA to conduct a more comprehensive risk assessment by 2006. In the 37th session of CCFAC held in April 2005, ochratoxin A was considered a high priority item for evaluation by JECFA, with particular reference to ochratoxin A levels in cereals, exposure assessment, and effects of processing on residual levels in foods.
Legal limits in some countries
29. Legal limits of ochratoxin A have been set in a number of food commodities including cereals and cereal products, dried vine fruits, roasted and soluble coffee, wine, grape juice, and foods for infants and children by the European Commission under EC regulation 466/2001 22 , 472/2002 23, 24 , and 123/2005 25 . The updated limits set for different food items are listed in Annex 1.
Study on ochratoxin A in food
30. Studies on levels of ochratoxin A in food, so far, have been conducted mainly in the West. Consequentially, international data accumulated at present are confined principally to the Western diet. Little is known about levels of ochratoxin A with regards to the rice-based Eastern diet pertaining to the weather conditions in countries in the East. The present study was carried out to evaluate the local situation of the levels of ochratoxin A in food, and to estimate the dietary exposure to ochratoxin A in secondary school students population in Hong Kong in order to assess if there is any associated health risk.
31. In this study, the potentials for any risks to health posed by ochratoxin A exposure from food intake are assessed by (1) comparing the prevailing levels of ochratoxin A in Hong Kong with reported levels in the international community and to the draft limit of 5 μg/kg proposed by Codex, and (2) comparing the dietary exposure to ochratoxin A with the appropriate safe exposure levels, the Provisional Tolerable Weekly Intakes (PTWIs), recommended by the Joint FAO/WHO Expert Committee on Food Additives (JECFA).
SCOPE OF STUDY
32. To study the prevailing levels of ochratoxin A in the Hong Kong retail market, this study covers major food commodities associated with ochratoxin A contamination including (i) cereals and cereal products, (ii) legumes, pulses and products (iii) meat, poultry and products (including offals), (iv) chocolate and cocoa products, (v) dried fruits, (vi) juice drinks, (vii) coffee and tea, and (viii) spices and condiments.
33. In order to assess the dietary exposure of secondary school student to ochratoxin A, the food items used for exposure assessment were based on those covered in the Food Consumption Survey in Secondary School Students carried out by FEHD in 2000 including the following food groups: (i) cereals and cereal products, (ii) legumes, pulses and products (iii) meat, poultry and products (including offals), (iv) chocolate, (v) dried fruits, (vi) juice drinks, and (vii) coffee and tea. In the FEHD Food Consumption Survey 2000, no individual consumption data is available for spices. It is expected that part of the dietary exposure to ochratoxin A due to spices may be included in the seasoned or marinated food items in this study.
34. This study did not cover food sources which are not associated with ochratoxin A contamination such as seafood. Products from ruminant animals (beef and milk) were not covered because they are generally not subject to the effects of ochratoxin A.
METHODOLOGY
Sampling Plan
35. Food samples were taken from the food groups cereals and cereal products, legumes, pulses and products, meat, poultry and products (including offals), chocolate and cocoa products, dried fruits, juice drinks, coffee and tea, as well as local spices and condiments. Food items that are sold loose or pre-packed were included in the study. All samples in the study were analysed on an individual basis.
36. Food samples were purchased from different premises in the local market, including supermarkets and fresh provision shops, in different locations in Hong Kong according to the food groups as mentioned in paragraph 32 (Scope of study) and sent to the Food Research Laboratory (FRL) of the Centre for Food Safety for analyses.
Sample Processing
37. Samples were analysed raw or cooked. For samples used for exposure analyses, samples were processed "as consumed" in the normal edible forms, which may involve cooking or after some forms of preparation as appropriate for the respective food items.
Laboratory analysis
38. Laboratory analysis for the detection and quantification of ochratoxin A was conducted by FRL using the method of High Performance Liquid Chromatography Tandem Mass Spectrometry (HPLC-MS/MS), using ochratoxin B (OTB) as an internal standard. All samples underwent processing steps of extraction, sample cleanup by immunoaffinity column and pre-concentration before submitting to HPLC-MS/MS for analyses of ochratoxin A levels. The limits of detection (LOD) achieved for solid and liquid samples were 0.05 μg/kg and 0.005 μg/kg respectively. These values compare favourably with LODs reported in overseas countries.
Data Interpretation
39. When the analytical value was below the LOD, the true value could be anywhere between zero and the LOD. In this study, the value of 1/2-LOD was assigned to all test results below LOD for the calculation of mean ochratoxin A levels in food commodities and for estimating the dietary exposure. Since the level of contaminants in food usually follows a log-normal distribution, assigning a value of 1/2 LOD to all non-detected levels is considered a conservative approach.
Dietary Exposure Assessment
40. Dietary exposures from individual food items were obtained based on the food consumption data (see paragraph 43) and the concentrations of ochratoxin A present in food.
41. Daily dietary exposure was multiplied by seven to obtain the weekly exposure level. Total exposure for each student was obtained by summing exposures from all food items. The mean and 95th percentile of the weekly exposure levels were used to represent the average dietary exposure and the exposure for high consumers respectively.
42. The estimated weekly exposure levels were then compared with the PTWI as established by JECFA.
Food Consumption Data
43. The food consumption data in this report were extracted from the Food Consumption Survey conducted in local secondary school students in 2000 by FEHD. In the survey, a stratified three-stage sampling plan was used, with a sampling frame of 472 secondary schools and more than 380,000 students, covering almost all the local secondary schools. A total of 967 students from 27 schools participated in the survey yielding a response rate of 77% at the school level and 96% at the student level. The mean weight of the participated students was 52.0kg 26 .
RESULTS
Levels of occurrence of ochratoxin A in the local market in Hong Kong
44. A total of 287 food samples were collected and sent to FRL for ochratoxin A analyses. 320 analyses were carried out for raw and cooked food items. The results for the prevailing levels of ochratoxin A in foods available in the local market in Hong Kong are shown in Annex 2.
45. Of the 287 food samples tested, ochratoxin A was reported in 101 items (35%). The measured concentration of ochratoxin A ranged from 0.01 to 2.09 μg/kg. 65% of the samples were below LOD. In particular, measurements for all 37 samples (64 analyses for uncooked and cooked items) in the group "rice and rice products" were all below LOD.
Exposure Analysis
Food Consumption Data
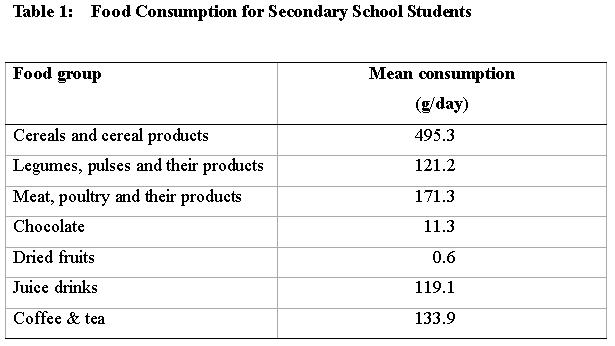
46. Food consumption data for the different food groups are given in Table 1.
Concentrations of Ochratoxin A in Food
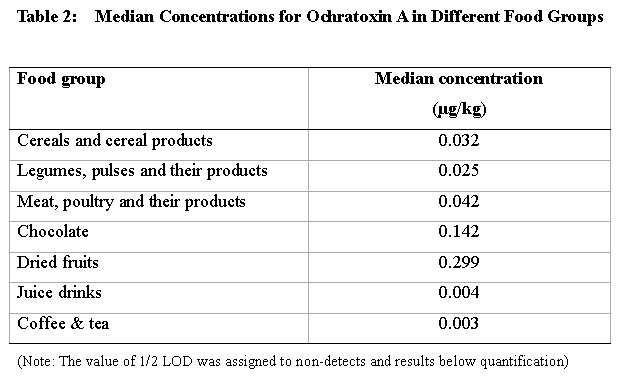
47. Food items used in exposure assessment were based on availability of Food Consumption data in the Food Consumption Survey 2000 for secondary school students. The median value for ochratoxin A concentration for each food group is shown in Table 2.
Dietary Exposures to Ochratoxin A
Average Secondary School Students
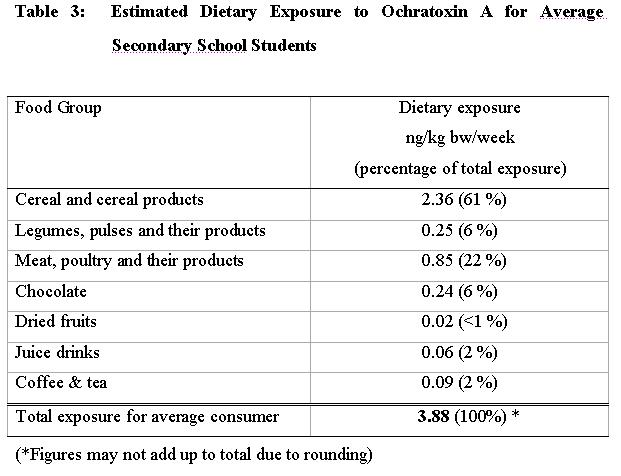
48. The dietary exposure to ochratoxin A for average secondary school students was estimated to be 3.88 ng/kg bw/week. The main dietary source of ochratoxin A was cereal and cereal products which contributed 61 % of the total exposure. This was followed by "Meat, poultry and their products" which contributed 22 %. Dietary exposures to ochratoxin A from different food groups are shown in Table 3.
High Consumers
49. Further analysis was undertaken to estimate the exposure level for high consumers. The 95th percentile exposure level of the secondary school students was used to represent the dietary exposure to ochratoxin A for high consumers and was estimated to be 8.97 ng/kg bw/week .
Comparison with PTWI
50. Dietary exposures to ochratoxin A for the average secondary school students and the high consumers were compared with the PTWI established by JECFA (Table 4).
DISCUSSION
Levels of ochratoxin A
51. The concentration levels of ochratoxin A in the raw cereal and cereal products tested in this study were all below the draft standard of 5 μg/kg proposed by Codex for raw cereals. Even with the low LOD levels of 0.05 μg/kg achieved in the FRL, only 35 % of all the food items tested in this study were detected with ochratoxin A levels. In particular, none of the food items in the subgroup "rice and rice products", which are staple food items with high consumption level in the local diet, had measurable levels of ochratoxin A. Ochratoxin A levels of <1 μg/kg in rice flour and rice-based cereals in Canada 27 and Italy 28 , and levels of 0.9 to 6.0 μg/kg in rice in Korea have been reported 29 . A recent study carried out in Taiwan in 2005 30 also reported no measurable ochratoxin A level in rice and rice products in 75 samples tested based on the limit of detection of 0.3 μg/kg.
52. The concentrations of ochratoxin A in all other food items in other food groups were low and were all below the 5 μg/kg level.
53. The reported levels of ochratoxin A in all food items tested in our study were also below the legal limits for the respective commodities in the European Union and other countries.
54. The low levels of ochratoxin A reported in this study indicated that the qualities of food commodities available in the retail market in Hong Kong are good. The low incidence in ochratoxin A contamination suggested a low rate of infection of the ochratoxin- producing fungi with subsequent production of toxin which may be due to good storage and weather conditions in our food supply.
Dietary Exposure
55. The dietary exposure to ochratoxin A for an average secondary school student was estimated to be 3.88 ng/kg bw/week while that for the high consumer was 8.97 ng/kg bw/week, amounting to 4% and 9% of the PTWI. Exposures to ochratoxin A for an average student and high consumer were both well below the PTWI established by JECFA of 100 ng/kg bw/week. These values were also below the tolerable daily intake established by the European Commission Scientific Committee for Food (ECSCF) of 5 ng/kg bw/day. Exposure estimates in the range 0.91 to 32.2 ng/kg bw per week were reported in the European Union. In Korea, an estimated probable daily intake for consumers has been reported to be in the range of 0.8 – 4.1 ng/kg bw/day. Exposure estimates for the Hong Kong secondary school student were in the lower ends of these levels reported in other countries. The low dietary exposure to ochratoxin A reported in this study indicates that it is unlikely that food commodities available in the retail market in Hong Kong would pose adverse health risk to the consumers with respect to ochratoxin A toxicity.
Contributions from Different Food Groups to Total Dietary Exposure
56. In our study, the main dietary source of ochratoxin A was "cereals and cereal products", which contributed to 61% of the total exposure. This is consistent with international data where cereals and cereal products were commonly reported as the major source of ochratoxin A exposure. Collated exposure data in the European Union (SCOOP task 3.2.7) reported a 50% contribution of the group "cereals and cereal products" to the total exposure. Risk assessment of ochratoxin A in the Netherlands reported that 55% of the total ochratoxin A intake from the diet was from cereals 31,. Despite the high proportion of the dietary intake of ochratoxin A due to cereals, the actual exposure to ochratoxin A through the consumption of cereals and cereal products in Hong Kong is low amounting to a mere 2.36 ng/kg bw/week.
57. "Meat, poultry and their products" ranked second accounting for 22% in the contribution to the total exposure of ochratoxin A. It is also the second highest food group for consumption level for secondary school student (Table 1). The exposure level due to this group is low amounting to only 0.85 ng/kg bw/week (Table 3). Food items within the group "meat, poultry and their products" would also have incorporated spices and condiments in their processing. Therefore, the exposure level due to "Meat, poultry and their products" included part of the ochratoxin A exposure due to spices and condiments which would augment the contributions and the amount of ochratoxin A exposure due to meat sources.
58. In the survey for the level of ochratoxin A occurrence, low levels of ochratoxin A contamination are reported for the group "spices and condiments". Survey results reported low ochratoxin A contamination in 16 out of 52 samples (31%) tested, all with levels below 1 μg/kg . The concentrations of ochratoxin A in the local spices (<0.05 to 0.82 μg/kg) and condiments (<0.005 to 0.99 μg/kg) were low compared with international data. In 2002, SCOOP task 3.2.7 reported an average ochratoxin A level of 1.15 μg/kg (<0.1 to 23.8 μg/kg) in the food group "spices" among member countries. A survey on spices carried out by Food Standard Agency of the United Kingdom (UKFSA) in 2005 reported a range of 0.3 to 47.7 μg/kg of ochratoxin A levels for chilli powder, cayenne pepper and paprika 32 .
59. In this study, separate consumption data for spices is not available from the food consumption data source, "FEHD Food Consumption Survey 2000". In order to estimate dietary exposure of ochratoxin A due to spices, the consumption levels for spices used by the SCOOP task 3.2.7 (Italian food consumption data) were used as a surrogate value, i.e. 2.1 g and 6.5 g per person per day for the average and high (95th percentile) consumer respectively. Taking this consumption data and the average ochratoxin A level of 0.17 μg/kg in spices obtained in our current study, it is estimated that an exposure of 0.05 ng/kg bw/week and 0.15 ng/kg bw/week may be expected for an average and high consumer respectively (corresponding to 1% and 4% of the estimated total dietary exposure to ochratoxin A, and 0.05% and 0.15% of PTWI, for an average and high consumer respectively). These values suggested that Hong Kong secondary school student may take in only low level of ochratoxin A from local spices.
Preventive Measures
60. Since ochratoxin A is stable and generally resistant to heat and processing, control of ochratoxin A contamination lies in the control of the growth of the toxin-producing fungi. Effective prevention of ochratoxin A contamination therefore depends on good farming and agricultural practices. Good Agricultural Practices (GAP) including methods to reduce fungal infection and growth during harvest, storage, transport and processing provide the primary line of defense against contamination of crops with ochratoxin A.
61. Specific measures to control the growth of ochratoxin A producing mould and the subsequent production of the toxin include lowering the moisture content of the grain at pre-harvest and harvest stages, employing effective drying processes, and ensuring dry conditions during subsequent storage and transport of the crops.
62. Extra precautions are required when conditions such as frost damage and excessive rainfall occur during preharvest, which may affect levels of ochratoxin A in the harvested grains.
63. Preservation of the grains is an important process in the prevention and reduction of ochratoxin A in cereals. Grain should be allowed to dry as much as possible before harvest. Ideally, grains are best harvested at a water activity below 0.70 (less than 14% moisture content in small grains). When it is not possible (e.g. adverse weather conditions leading to a disrupted harvest with the grain harvested at high moisture contents), grain should be dried to a moisture content corresponding to a water activity of less than 0.70 as quickly as possible immediately after harvest and preferably using heated-air drying.
64. Other general measures for the prevention of mycotoxin contaminations include maintainence of good soil conditions and plant nutrition, prevention of fungal contaminations during planting, minimising insect or mechanical damage during pre-harvest and harvest stages, and avoiding sun-drying of food commodities at high humidity which are prone to fungal infections.
65. Detailed measures and practices based on Good Agricultural Practices (GAP) and Good Manufacturing Practices (GMP) for the prevention and reduction of contamination by ochratoxin A in cereal grains may be obtained in the "Code of practice for the Prevention and Reduction of Mycotoxin Contamination in Cereals, including Annexes on Ochratoxin A, Zearalenone, Fumonisins and Tricothecenes" laid out by Codex Alimentarius Commission in 2003. In addition, an "Ochratoxin A Management System based on Hazard Analysis Critical Control Points (HACCP)" has also been put forward in this Codex document (CAC/RCP 51, 2003) 33.
LIMITATIONS
Food Consumption Survey for Secondary School Students 2000
66. In the food consumption survey, the food consumption pattern of secondary school students was collected using a food frequency questionnaire. Although the food frequency questionnaire used was very comprehensive, it was not possible to cover every single food item, some of which might be relevant to ochratoxin A exposure. FEHD is currently conducting a more comprehensive population-based food consumption survey which would allow more accurate estimates and more comprehensive exposure studies to be conducted in the future. In our study, consumption levels for coffee and tea were grouped and consumption data for spices is not available in our food consumption survey.
Sampling size
67. For exposure analyses, at least three samples for each food item were taken from the retail market in this study. A more precise estimate of the mean ochratoxin A concentration for each particular food item may be obtained by increasing the number of samples taken for each food item for laboratory analysis. However, the number of samples taken has to be balanced with the required resources and the number of food items to be included.
CONCLUSIONS AND RECOMMENDATIONS
68. Of 287 food samples tested, measurable amounts of ochratoxin A were found present in 101 food samples tested which account for 35% of all samples. All the 101 samples were reported to have low levels of ochratoxin A ranging from 0.01 to 2.09 μg/kg. These values were below the proposed Codex draft maximum level of 5 μg/kg. This study showed that ochratoxin A levels in cereals and cereal products and other commonly consumed food available in the retail market in Hong Kong were low. In particular, no measurable amount of ochratoxin A was found in our local staple foods of rice and rice products.
69. Dietary exposures to ochratoxin A were estimated to be 3.88 and 8.97 ng/kg body weight/week for average secondary school student and high consumers respectively. These values were far below the provisional tolerable weekly intake (PTWI) of 100 ng/kg body weight established by JECFA in 2001. It can be concluded that secondary school students are unlikely to experience major toxicological effects of ochratoxin A.
Advice to trade
70. To minimise ochratoxin A contamination in foods, the trade is advised to:
- follow Good Agricultural Practices (GAP) and Good Manufacturing Practices (GMP) throughout the stages of planting, pre-harvest, harvest, storage, transport, processing and distribution of food commodities;
- develop ochratoxin A management system based on Hazard Analysis Critical Control Points (HACCP) for food production;
- obtain raw materials from reliable and reputable suppliers;
- maintain good storage conditions:
- store foods in cool and dry environment
- keep stocks according to a first-in-first-out basis
- keep proper documentations of control points.
Advice to consumers
71. The present study showed that the prevailing levels of ochratoxin A in food available in the local market in Hong Kong are low and does not warrant a change in the dietary pattern for the public. Members of the public are advised to follow the general dietary advice of healthy eating and to note the following:
- consume a variety of cereals as the basic requirement of a healthy diet;
- have a balanced and varied diet, do not overindulge in a small variety of food items;
- take note of the conditions of the food products before purchase;
- do not purchase or consume foods with the presence of visible moulds, or foods that are dampened with unexpected moistures;
- purchase foods from reliable and reputable retailers;
- store foods in a cool, dry place before consumption and according to storage instructions;
- avoid stocking up excessive foods and take notice of the durability of foods.
REFERENCES
- Van der Merwe, K.J., Steyn, P.S., and Fourie, L., Scott, D.B., and Theron, J.J. (1965). Ochratoxin A , a toxic metabolite produced by Aspergillus ochraceus Wilh. Nature 205 (4976): 1112
- Van der Merwe, K.J., Steyn, P.S., and Fourie, L., Mycotoxins. Part II. The constitution of ochratoxin A, B, C, metabolites of Aspergillus ochraceus Wilh. Journal of the Chemical Society 7038: 1965.
- Li, S., Marquardt, R.R, Frohlich, A.A., Vitti, T.G., and Crow, G. (1997). Pharmacokinetics of ochratoxin A and its metabolites in rats. Toxicology and Applied Pharmacology 145:82.
- European Commission. Assessment of dietary intake of ochratoxin A by the population of EU Member States, (1997) SCOOP. Task 3.2.2. Reports on tasks for scientific cooperation. Report EUR 17523.
- European Commission. Reports on tasks for scientific cooperation. (2002) Reports of experts participating in Task 3.2.7. Assessment of dietary intake of ochratoxin A by the population of EU Member States, SCOOP Task 3.2.7, January, 2002.
- Kiessling, K.H., Pettersson, H., Sandholm, K. & Olsen, M. (1984) Metabolism of aflatoxin, ochratoxin, zearalenone, and three tricothecenes by intact rumen fluid, rumen protozoa, and rumen bacteria. Appl. Environ. Mircobiol., 47, 1070-1073.
- European Mycotoxin Awareness Network. Fact Sheet 3. Ochratoxin A. Available from: URL: http://193.132.193.215/eman2/fsheet3.asp
- International Program of Chemical Safety. Selected Mycotoxins: Ochratoxins, tricothecenes, ergot. Environmental Health Criteria 105. World Health Organisation, Geneva, 1990. Available from: URL: http://www.inchem.org/documents/ehc/ehc/ehc105.htm
- Berry, L. (1988) The pathology of mycotoxins. Journal of Pathology 154: 301
- Puntaric, D., Bosnir, J., Smit, Z., and Baklaic, Z. (2001). Ochratoxin A in corn and wheat: Geographical association with endemic nephropathy. Croatian Medical Journal. 42(2):175.
- Codex alimentarius commission. Position Paper on Ochratoxin A. CCFAC Agenda item 14(a) CX/FAC 99/14. November 1998
- Galtier, P. (1978) Contribution of pharmacokinetics studies to mycotoxicology – Ochratoxin A. Vet. Sci. Commun. 1, 349-358.
- Suzuki, S., Satoh, T. & Yamazaki, M . (1977) The pharmacokinetics of ochratoxinA in rats. Jpn. J. Pharmacol., 27, 735-744.
- Galtier, P., Alvinerie, M. & Charpenteau, J.L. (1981) The pharmacokinetic profiles of ochratoxin A in pigs, rabbits and chickens. Food Cosmet. Toxicol., 19, 735-738.
- Harwig, J., Kuiper-Goodman, T. & Scott, P.M. (1983) Microbial food toxicants: Ochratoxins. In: Rechcigl, M., ed., Handbook of Foodbourne Diseases of Biological Origin, Boca Raton, FL: CRC Press, pp. 193-238.
- Albassam, MA., Yong, S.I., Bhatnagar, R., Sharma, A.K. & Prior, M.G. (1987) Histopathological and electron microscopic studies on the acute toxicity of ochratoxin A in rats. Vet. Pathol., 424, 427-435.
- JECFA. Ochratoxin A (JECFA 47, 2001) Available from: http://www.inchem.org/documents/jecfa/jecmono/v47je04.htm
- Creppy EE, Rosechenthaler R & Dirheimer G, (1984). Inhibition of protein synthesis in mice by ochratoxin A and its prevention by phenylalanine. Food. Chem. Toxicol., 22, 883-886.
- IARC (1993) Ochratoxin A. In: IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Humans, Vol.56, Lyon: IARC Press, pp. 489 – 521.
- JECFA (2002) Evaluation of Certain Mycotoxins in Food. (56th Report of the Joint FAO/WHO Expert Committee on Food Additives). Technical Reports Series No. 906, 2002.
- EUROPA European Commission Research. Prevention of ochratoxin A in cereals. Dr. Monica Olsen. Available from URL: http://europa.eu.int/comm/research/quality-of-life/ka1/volume1/qlk1-1999-00433.htm.
- European Commission. Commission Regulation (EC) No 466/2001 of 8 March 2001. Official Journal of the European Communities No. L77 1-13.
- European Commission. Commission Regulation (EC) No 472/2002 of 12 March 2001. Official Journal of the European Communities No. L75, 18-20.
- European Commission. Corrigendum of 23 March 2002. Official Journal of the European Communities No. L80, 42.
- European Commission. Commission Regulation (EC) No 123/2005 of 26 January 2005 amending Regulation (EC) No 466/2001 as regards ochratoxin A. Official Journal of the European Union No. L25, 3-5
- FEHD. Food Consumption Survey 2000. Hong Kong Food and Environmental Hygiene Department; 2001.
- Lombaert GA, Pellaers P, Roscoe V, Mankotia M, Neil R, Scott PM (2003) Mycotoxins in infant cereal foods from the Canadian retail market. Food Addit Contam. 2003, 20, 494-504
- Biffi R, Munari M, Dioguardi L, Ballabio C, Cattaneo A, Galli CL, Restani P. (2004) Ochratoxin A in conventional and organic cereal derivatives: A survey of the Italian market, 2001-02. Food Addit Contam., 21, 586-591
- Park JW, Chung S-H, and Kim Y-B. (2005). Ochratoxin A in Korean Food Commodities: Occurrence and Safety Evaluation. J. Agri. Food Chem. (53) 4637 – 4642.
- Lin L-C, Chen P-C, Fu Y-M and Shih D Y-C. (2005) Ochratoxin A Contamination in Coffees, Cereals, Red Wines and Beers in Taiwan. 13 (1), 84 -92.
- Baker M, Pieters MN. (2002) Risk Assessment of ochratoxin A in the Netherlands. RIVM report 388802025/2002.
- UKFSA. 2005. Survey of Spices for Aflatoxins and Ochratoxin A. Food Survey Information Sheet. Available at URL: http://www.food.gov.uk/science/surveillance/fsis2005/fsis7305
- Codex Alimentarius Commission. CAC/RCP 51, 2003 Prevention and Reduction of Mycotoxin Contamination in Cereals, including Annexes on Ochratoxin A, Zearalenone, Fumonisins and tricothecenes. Available at URL: http://www.codexalimentarius.net/web/standard_list.jsp
Annex 1
Legal limits for ochratoxin A in different food products set by the European Commission: (For details, please refer to Commission Regulation (EC) No.123/2005 of 26 January 2005)