
Food Safety Focus (39th Issue, October 2009) – Incident in Focus
Ethyl Carbamate and Alcohol
Reported by Dr. Anna S.P. TANG, Scientific Officer,
Risk Communication Section, Centre for Food Safety
Last month, the Centre for Food Safety (CFS) released the results of a study which examined the level of ethyl carbamate (EC) in local fermented foods and beverages. The study found that EC was present in varying amounts in different local fermented food and beverage items. In some alcoholic beverages such as yellow wine, sake and plum wine, relatively higher levels of EC were found. As a group, "alcoholic beverages" was identified as the main dietary source of EC for alcohol drinkers. In this article, we will talk more about alcohol in relation to EC.
What is EC?
Ethyl carbamate also known as urethane, is a contaminant naturally formed in fermented foods and alcoholic beverages during the fermentation process or during storage. EC has also been used as human medicine which is now banned due to toxicological concerns and lack of effi cacy. Variable levels of EC have been found in different alcoholic beverages (Figure 1). For example, spirits, particularly those made from stone-fruits such as cherries, apricots or plums, usually contain higher amount of EC while beers contain low levels. EC levels can differ greatly between different types of alcoholic beverages e.g. the mean EC contents in fruit brandy in some reports were over 600 times that of beer.
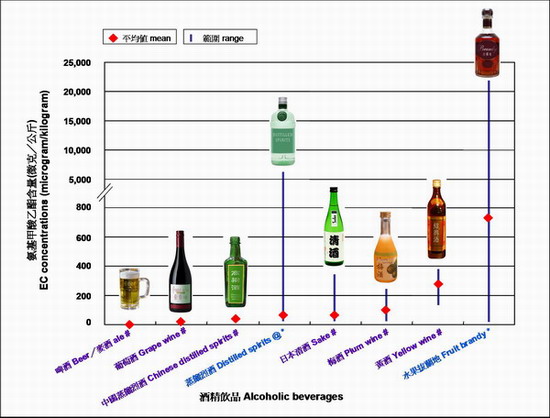
Figure 1. Ethyl carbamate (EC) levels in various types of alcoholic beverages
# Levels of EC drawn from a limited number of samples tested in the risk assessment study on Ethyl Carbamate in Local Fermented Foods conducted by the CFS
* Levels of EC reported overseas (WHO Food and Nutrition Paper 82, 2006; EFSA, 2007)
@ Distilled spirits excluding fruit brandy
Formation of EC in Alcoholic Beverages
Ethyl carbamate can be formed from various substances present in alcoholic beverages and their break-down products as a result of the fermentation process. These precursor substances, e.g. urea, cyanate and citrulline, react with ethanol to form EC in alcoholic beverages. The amount of EC formed depends on the key factors of light exposure and elevated temperature.
Health Effects of EC
Public concerns regarding EC in foods are related to its potential to cause cancer in human. EC has been observed to cause various types of cancer in experimental animals. In 2007, the International Agency for Research on Cancer (IARC) up-graded the classification of EC to Group 2A "probably carcinogenic to humans".
The Joint Food and Agriculture Organization/World Health Organization Expert Committee on Food Additives (JECFA) evaluated EC in 2005. It concluded that intake of EC from foods excluding alcoholic beverages would be of low concern. However, dietary exposure to EC from both food and alcoholic beverage was of concern and measures to reduce concentrations of EC in some alcoholic beverages were recommended.
Key Points to Note
- Public concerns regarding EC in foods are related to its potential to cause cancer in human.
- Formation of EC in alcoholic beverages can be reduced significantly by keeping them at low light conditions and at lower temperatures e.g. at or below 20°C.
- Members of the public should take a balanced diet and avoid overindulgence of alcoholic beverages.
Local Dietary Exposure to EC
A risk assessment was conducted by the CFS based on the EC levels measured in our study and the preliminary consumption data of the Population based Food Consumption Survey 2005 - 2007. It was found that for the general population, dietary exposure to EC from consumption of fermented foods and beverages is unlikely to pose health concerns. However, health risk of EC cannot be ruled out for high consumers of alcoholic beverages, e.g. distilled spirits (>270mL/day), plum wine (>76mL/day), grape wine (>250mL/day) on a regular basis.
Reduce EC Levels in Alcoholic Beverages
Formation of EC can be reduced significantly when alcoholic beverages are kept at low light conditions and at lower temperatures. During transport and storage, care should be taken to maintain the correct cold chain preferably at or below 20°C and critically not above 38°C. At the temperatures at which most wine cellars are maintained to keep the sensory properties and qualities of grape wine (i.e. below 20°C ), less increment in EC content is expected.
The CFS has worked closely with the trade to devise the Guidelines on reducing the level of ethyl carbamate in alcoholic beverages during storage and transport which are applicable to importers, distributors, wholesalers and retailers of alcoholic beverages. Members of the trade should make efforts to reduce the level of EC in alcoholic beverages.
Advice to the Trade
- Manufacturers should follow Good Manufacturing Practice (GMP). Develop mitigation measures to reduce the levels of EC in alcoholic beverages, e.g. identifying and reducing the amount of precursors.
- Use proper containers to protect alcoholic beverages from light exposure.
- Importers, distributors, wholesalers and retailers should minimise heat and light exposure during transportation and storage of alcoholic beverages. Special care should be taken to maintain the correct cold chain preferably at or below 20°C, and critically not above 38°C.
- Obtain alcoholic beverages from reliable suppliers.
- Keep stock according to the first-in-first-out principle.
Advice to the Public
- Maintain a balanced diet. Avoid overindulgence of alcoholic beverages.
- Store alcoholic beverages in a cool place under low light conditions.
- Avoid stocking up excessive alcoholic beverages to minimise the duration of storage.

