Food Safety Focus (32nd Issue, March 2009) – Food Safety Platform
Nutrient and Health – Fats
Reported by Ms. Jacqueline FUNG, Scientific Officer
Risk Communication Section, Centre for Food Safety
Previously, we introduced protein and carbohydrates; in this issue, we will take a look at the last macronutrient – fat, which belongs to a class of nutrients called lipid.
We often refer triglyceride as fat in our daily life. In fact, ninety-eight percent of dietary fat is in the form of triglyceride. Triglyceride has a glycerol as the backbone with three fatty acids attached. Fatty acids exist in different chemical structures. Due to the uniqueness in structures, different fats exhibit different properties and in turn have dissimilar health effects.
Saturated, Monounsaturated or Polyunsaturated Fatty Acids
By the number of double bonds in fatty acids, we classify them as saturated, monounsaturated or polyunsaturated fatty acids. A saturated fatty acid (SFA) has no double bond, monounsaturated fatty acid (MUFA) has one double bond, whereas polyunsaturated fatty acid (PUFA) has two or more double bonds. It is believed that the number of double bonds in fatty acids is inversely related to the risk of cardiovascular diseases. As such, saturated fats, with no double bond, are associated with higher risk of heart diseases.
In general, most animal fats have higher percentage of saturated fatty acids and they tend to be in solid state at room temperatures. On the other hand, olive and canola oils are rich in monounsaturated fatty acids, whereas corn, soybean, peanut and fish oils are rich in polyunsaturated fatty acids. Fats that have higher percentage of mono- and/or polyunsaturated fatty acids (except those undergone hydrogenation) tend to be in liquid state at room temperatures.
Both essential fatty acids, α -linolenic acid and linoleic acid, are polyunsaturated fatty acids. Metabolism in the body produces longer chain fatty acids, such as docosahexaenoic acid (DHA) from α -linolenic acid and arachidonic acid (ARA) from linoleic acid.
Cis or Trans Fatty Acids
The term "cis" and "trans" describe the positions of the two hydrogen atoms located next to the carbon atoms where the double bond exists. Cis fatty acid has both hydrogen atoms located on the same side. On the contrary, trans fatty acid has the two hydrogen atoms on opposite sides. Fatty acids with cis configuration are typical in natural foods. Most of the trans fatty acids are formed during the process of hydrogenation of vegetable oils. Trans fatty acids are not essential and have no known health benefit. In fact, similar to saturated fatty acids, they may increase the risk of heart diseases. For more on trans fat, please look up previous Food Safety Focus articles on trans fat.
Functions and Intake of Fat
Fat is the concentrated source of energy (1g of fat = 9kcal). Besides, among many important bodily functions, fat is the carrier of fat-soluble vitamins (i.e. vitamin A, D, E and K) and is the structural components of cell membrane.
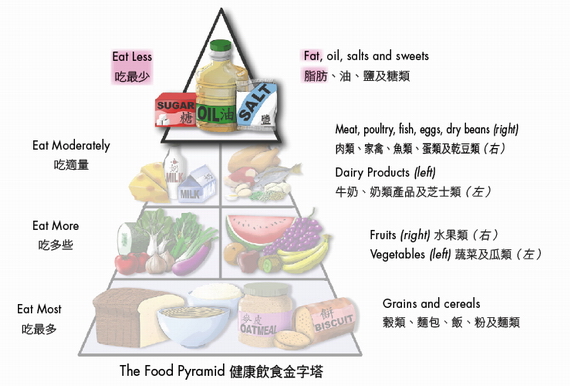
According to the Chinese Adequate Intake (AI), 20-30% of energy contribution should come from fats, which means that there should be about 44-67g of fat from all food sources in a 2 000-kcal diet. Referring to the food pyramid, we should "Eat Less" fats and oils (one teaspoon of oil weighs about 5g) by avoiding fatty meats and fried foods, plus selecting foods in lower fat version (e.g. a 250mL-serving of whole milk has a minimum of 8.75g of fat as compared to about 5g fat in low-fat milk with the same serving size.)
After talking about fat, next time we will examine another lipid – cholesterol.