Food Safety Focus (31st Issue, February 2009) – Food Safety Platform
Nutrient and Health - Carbohydrates: Sugars
Reported by Ms. Jacqueline FUNG, Scientific Officer,
Risk Communication Section,Centre for Food Safety
In the last issue, we focused on complex carbohydrates (i.e. starch and dietary fibres). In this issue, we will talk about simple carbohydrates (i.e. sugars).
Simple Carbohydrates (Sugars)
Based on the chemical structures, simple carbohydrates can be divided into two categories, namely monosaccharides (single sugar molecule) and disaccharides (two single sugar molecules joined together). Glucose, fructose and galactose are the three monosaccharides important in nutrition. These single sugar molecules contain 6 carbon atoms, 12 hydrogen atoms and 6 oxygen atoms (i.e. chemical formula as C6H12O6).
Among these three common monosaccharides, glucose is of most significance as it always exists as one of the two sugar molecules found in a disaccharide, plus it is the basic unit that makes up starch and glycogen. Furthermore, it is worth mentioning that our brain is almost exclusively dependent on glucose as energy source.
Fructose, having the same chemical formula as glucose, has different chemical structure. Because of its unique structure, it stimulates the taste buds which result in a sweet sensation. Fructose is the "sweetest" of all sugars (i.e. with the highest intensity of sweetness) and it is most abundant in fruits and honey.
On the contrary, galactose, the other monosaccharide, has low sweet intensity and can rarely be found free in foods. It combines with a glucose molecule to form the disaccharide lactose, commonly known as milk sugar, since it is found in milk (4.7% in cow's milk and 7% in human breast milk).
Another disaccharide that is not widely available in foods is maltose. Maltose is composed of two glucose molecules and it is produced when seeds (e.g. barley) germinate and starch breaks down during digestion.
The most familiar disaccharide of all is sucrose, which we usually call table sugar in our daily life. Sucrose is the combination of fructose and glucose. To produce table sugar, sucrose is extracted from the juices of sugar cane and sugar beets. Sucrose is widely used as an ingredient for sweetening purpose in food production. Besides, it is often used to improve the texture, structure and consistency of foods. In addition, it also functions as a preservative in jams and jellies.
Table: Simple Carbohydrates (Sugars)
| Monosaccharides | Disaccharides |
|---|---|
| Glucose | Sucrose (glucose + fructose) |
| Fructose | Lactose (glucose + galactose) |
| Galactose | Maltose (glucose + glucose) |
Sugars Intake
Most people love sweets, but excessive intake may increase the risk of obesity. Obese individuals are more prone to chronic diseases, such as diabetes, hypertension and heart diseases. According to the World Health Organization's recommendation, less than 10% of energy contribution should come from sugars added to foods during the production and consumption plus natural sugars found in honey, fruit juices and syrups. In other words, a 2 000-kcal diet should have less than 50g of sugars.
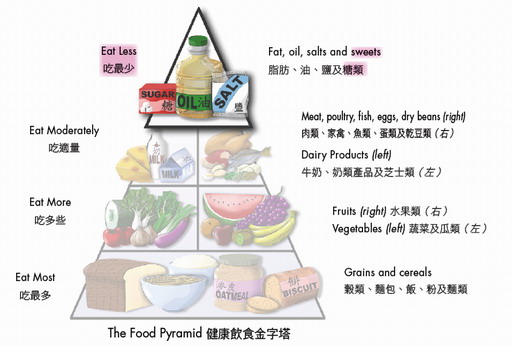
Illustration: The Food Pyramid
With reference to the food pyramid, it is the top of the pyramid which represents the "Eat Less" level, thereby intake of sugars should be kept at a low level. To do so, individuals can reduce the use of sugars/syrups in beverages (one level and one heaped teaspoon of sugars weigh about 4g and 6g respectively) and cut down the consumption of confectionaries, desserts and soft drinks (one can of soft drinks contains approximately 30g to 43g of sugars, i.e. close to a day's limit). To learn about the sugar contents of foods, please visit the Nutrient Information Inquiry System at the CFS's website .
Illustration: One heaped teaspoon of sugars ≈ 6g, one level teaspoon of sugars ≈ 4g
After protein and carbohydrates, we will continue the series on nutrition and introduce the last macronutrient - fats in the next issue.